35 KiB
Ion channels underlie action potentials
- Predictions about the nature of ion channels from Hodgkin and Huxley:
- Because conductances are large, channels must be able to pass ions at high rate
- Channels must be gated by the membrane potential
- Different channels for Na⁺ and K⁺
- Problem– Voltage clamping cannot look at individual channels...it's measuring the aggregate current flowing through a whole bunch of channels at once. What does an individual channel look like? How does it work?
- Solution– Patch Clamping
Note:
Today we will take a closer look at the nature of ion channels and how they are able to exhibit their remarkable properties that enable action potentials and all forms of electrical signaling in the nervous system.
Now we know from our previous classes covering the work by Hodgkin-Huxley, that there are some predictions we make concerning the nature of ion channels..
--
Question
During the rising phase of the action potential:
- a. All sodium channels are closed
- b. Some of the sodium channels are closed
- c. All potassium channels are open
- d. All sodium channels are open
- e. The membrane potential is hyperpolarizing
Note:
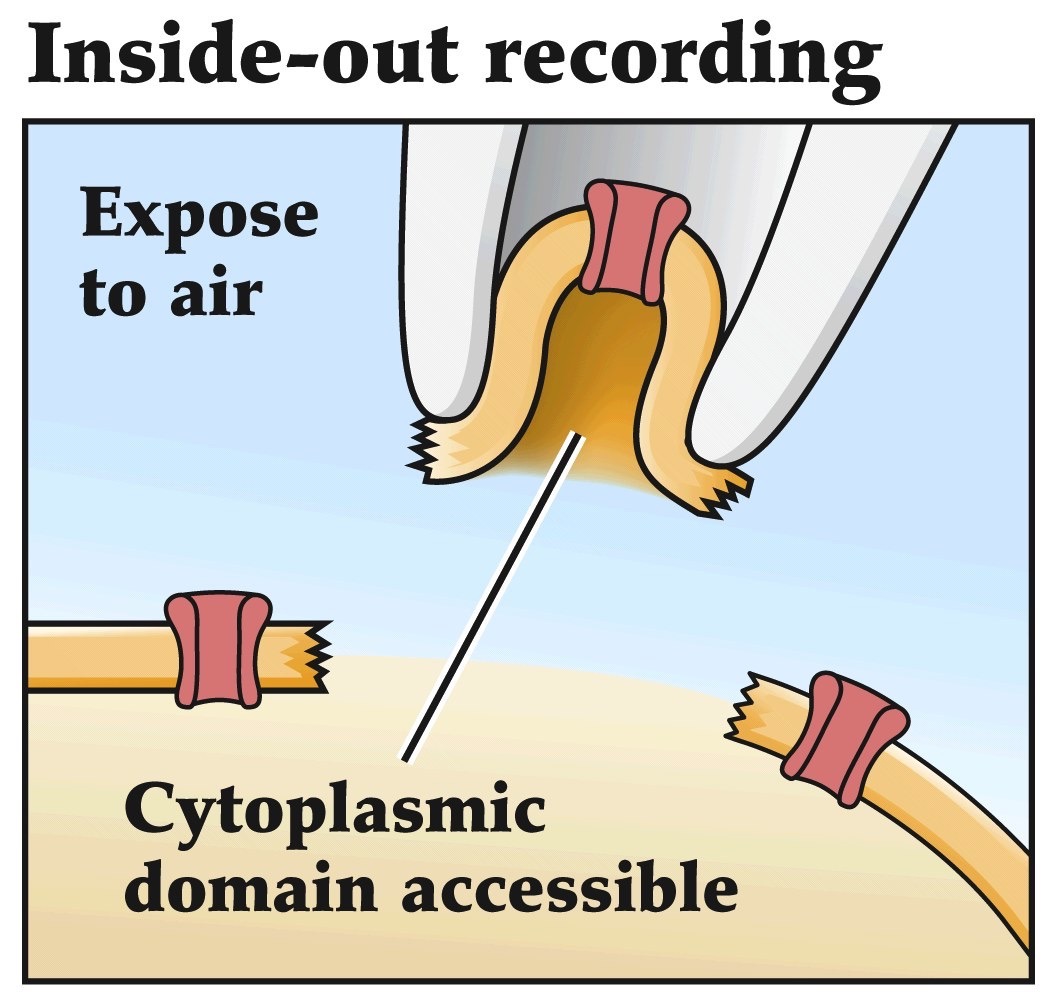
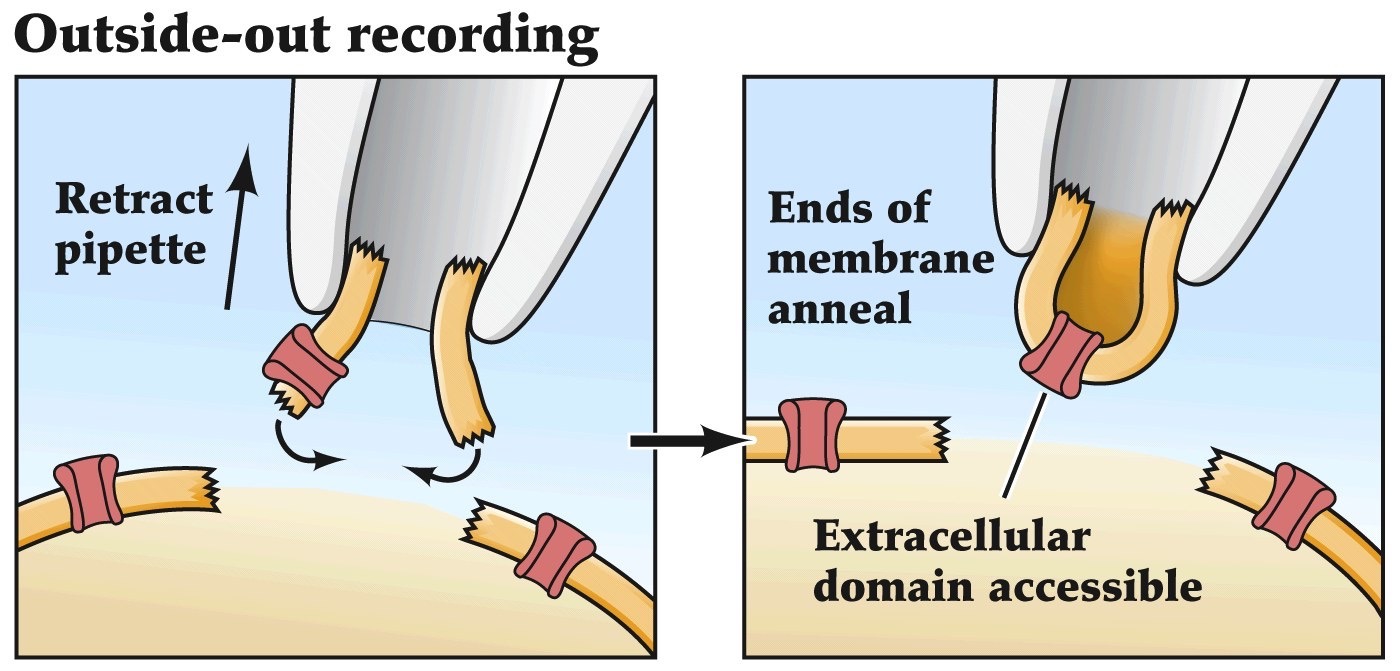
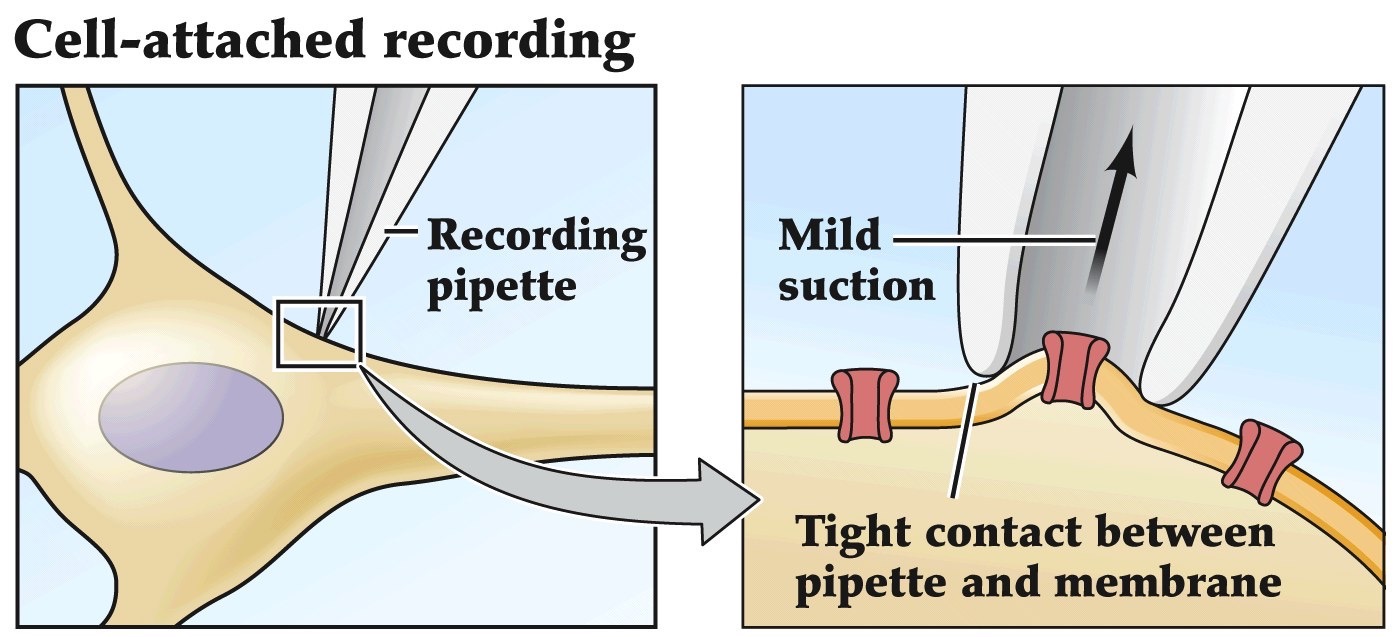
Patch clamp recordings
- Allows one to look at currents flowing through a single channel
- Pipette with small opening makes a tight seal with the membrane
- Currents are amplified and measured
- Can be adapted to do whole cell recordings, inside out recordings or outside out recordings
Note:
Remember voltage-clamp recordings that we've been talking about before. Patch-clamping is an adaptation of the voltage-clamp method that allows you to assess the currents flowing across very small patches of lipid membrane. So if you have one or few ion channels in that small patch of cell membrane you can study the microscopic functional properties of those individual channels.
The patch clamp method
Can measure ion flow through a single channel.
Note:
--
The patch clamp method
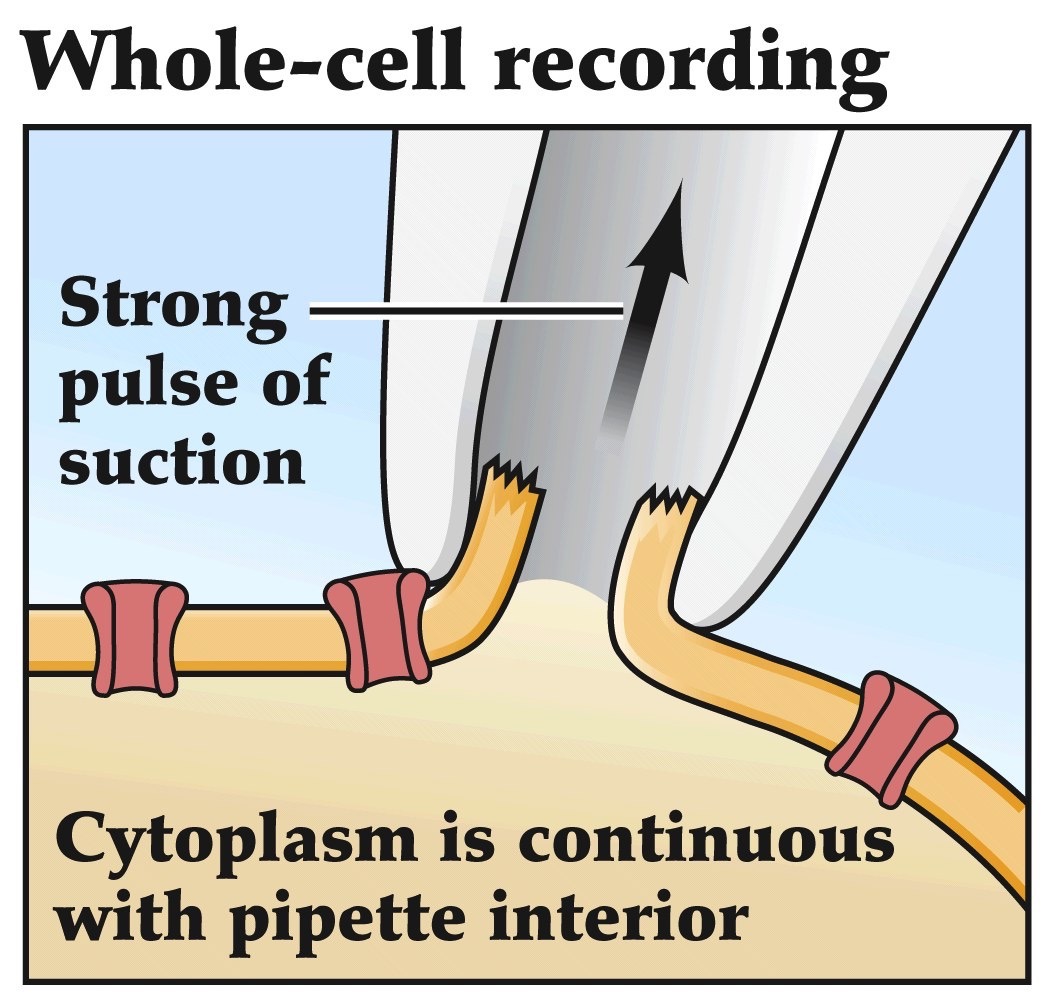
Can measure potentials and currents
from entire cell and introduce
things into the cytoplasm
Note:
--
The Nobel Prize in Physiology or Medicine (1991)
"for their discoveries concerning the the function of single ion channels in cells"
Note:
Patch clamp established by E. Neher adn B. Sakmann at Max Planck Institute in Germany 1976.
Patch clamping Na⁺ channels
- Block K⁺ channels with Cs⁺ or with tetraethylammonium (TEA)
- Brief depolarizations cause small inward currents that disappear right away
- Each inward current is the opening of one Na⁺ channel
- About 1-2 pA of current ≈ thousands of ions/ms
- Stochastic opening, biased at the beginning of a pulse
- Probability of opening varies with membrane potential
- If you remove Na⁺ from medium, do not see currents
- Tetrodotoxin (TTX) blocks currents
Note:
- TEA
- tetraethylammonium
- quaternary ammonium cation
- blocks voltage gated K+ channels
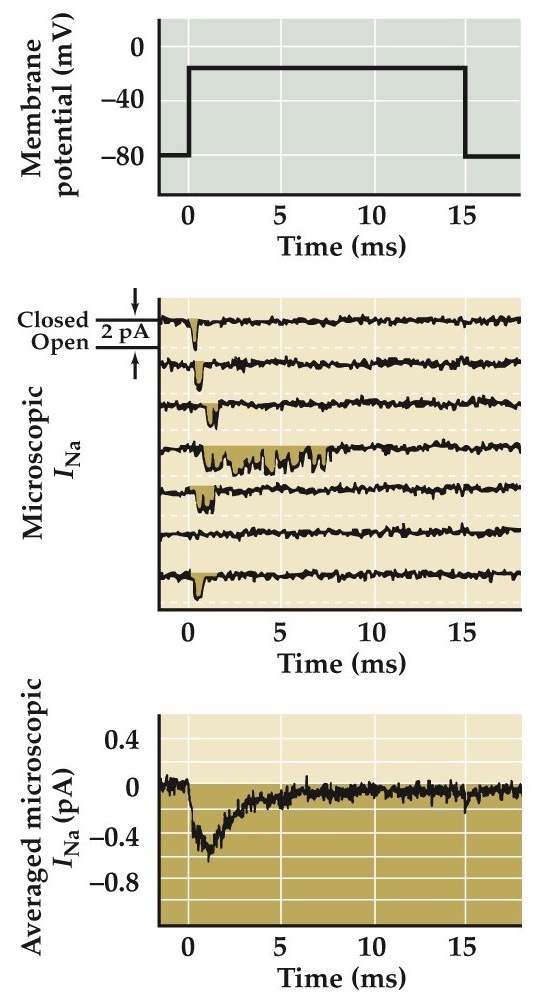
Measurements of ionic currents flowing through single Na⁺ channels
- Small (picoampere) inward currents
- Unitary amplitudes
- Open at beginning of pulse
- Inactivate quickly
Note:
Patch a piece of membrane and block K currents. Do a bunch of short recordings while clamping the membrane at depolarized potential. e.g. here is 7 experimental trials. Notice the amplitude is discrete— it is unitary. If you were recording from lots of these single channels simultaneously or added together all the recordings from one channel you'd -->
Transient channel opening in Na⁺ channels (inward current).
This research is from Bezanilla and Correa 1995, Vandenburg and Bezanilla 1991, Correa and Bezanilla 1994
- unitary (wn, adj)
- one, unitary -- (having the indivisible character of a unit; "a unitary action"; "spoke with one voice")
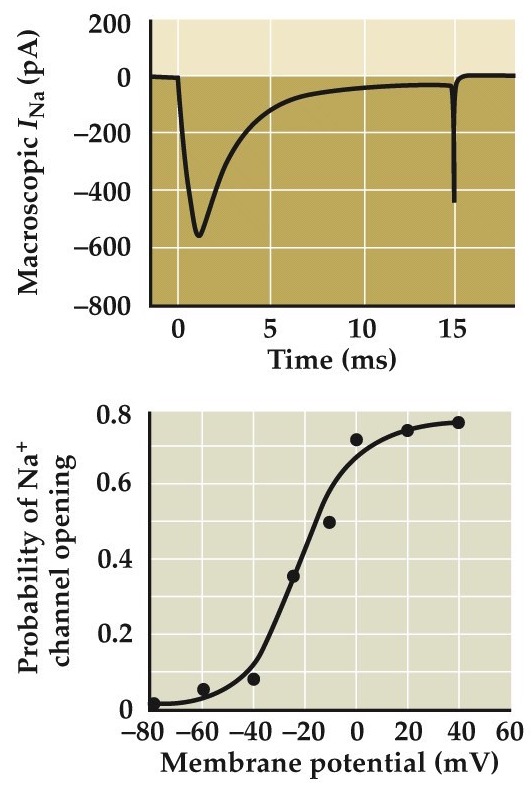
Measurements of ionic currents flowing through single Na⁺ channels
- Summed current from many single channels looks like macroscopic currents seen in voltage clamping
- Probability of opening increases as a function of membrane potential
Note:
Average the microscopic currents together and you get something very similar to this macroscopic voltage-clamp current shown at the top.
Notice that even at -20 to -10mV when you expect an action potential to be well into its rising phase above threshold, the probability of sodium channel opening is just 40-50% (and never reaches 100%).
Bezanilla and Correa 1995, Vandenburg and Bezanilla 1991, Correa and Bezanilla 1994
Patch clamping K⁺ channels
- Add tetrodotoxin (TTX) to block Na⁺ channels
- Depolarization pulses cause outward currents
- Once a channel opens (usually with a delay) it remains open for the duration of the pulse
- The probability of channel opening depends on the membrane potential
- Sensitive to TEA
Note:
Now let's examine patch clamp experimental data from K+ channels.
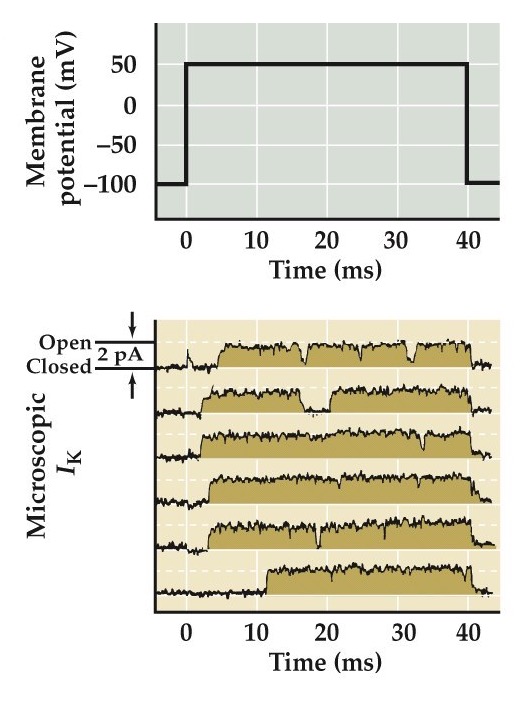
Measurements of ionic currents flowing through single K⁺ channels
- Early delay in opening
- Once open stay open
Note:
Sustained channel opening in K⁺ channels (outward current).
This research is from Augustine and Bezanilla, Hille 2001; Augustine and Bezanilla 1990; Perozo et al 1991
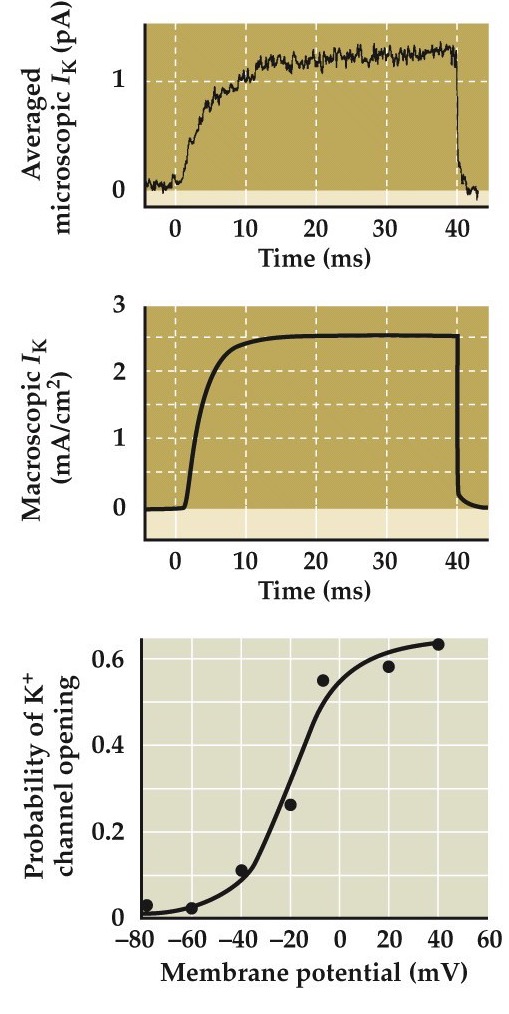
Measurements of ionic currents flowing through single K⁺ channels
- Summed current from many single channels looks like macroscopic currents seen in voltage clamping
- Probability of opening increases as a function of membrane potential
Note:
Sum a bunch of these microscopic channel currents and you get this top curve and which looks very similar to the macroscopic current curve as we’ve seen previously.
This research is from from Augustine and Bezanilla, Hille 2001; Augustine and Bezanilla 1990; Perozo et al 1991
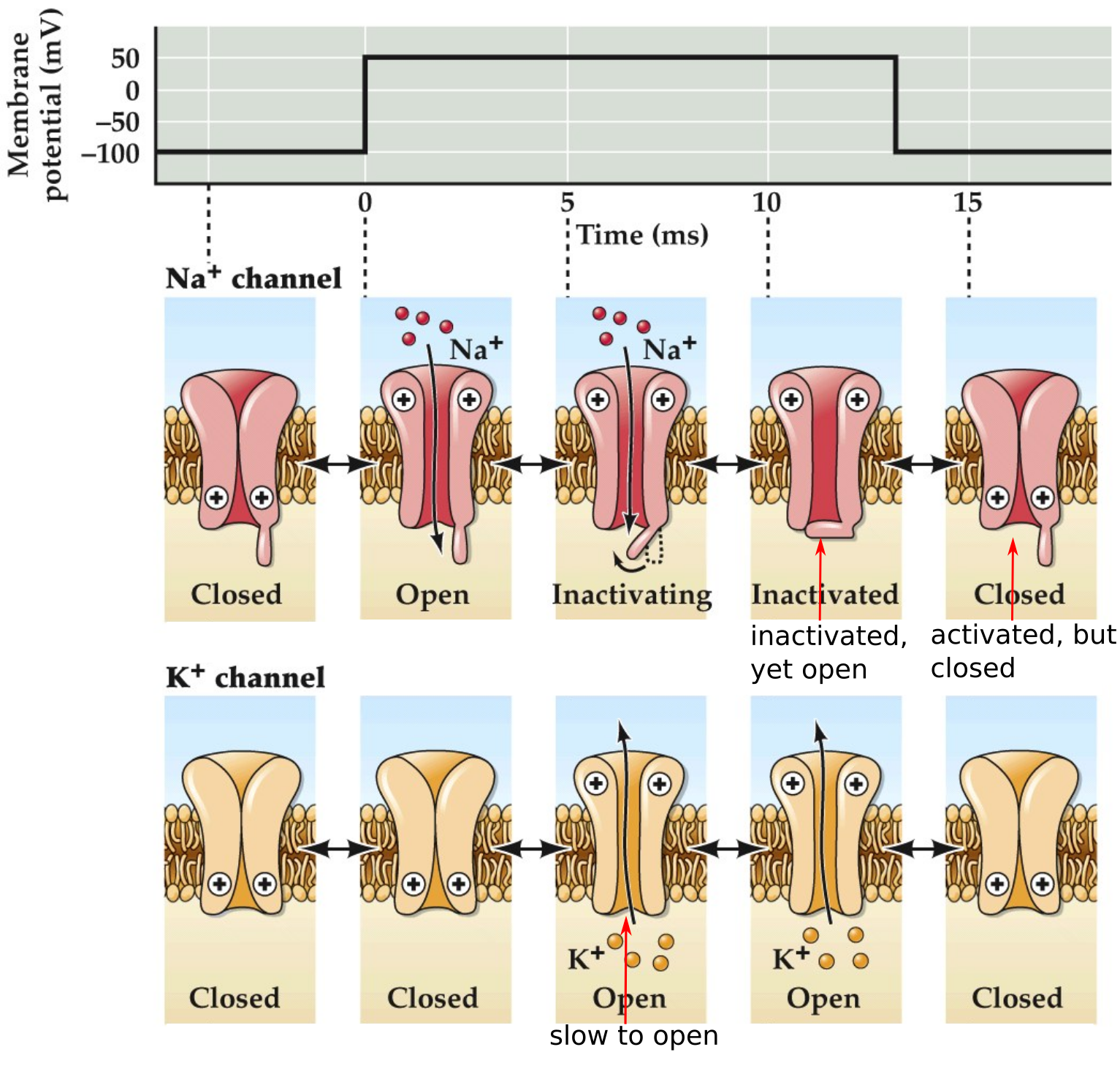
Functional states of voltage-gated Na⁺ and K⁺ channels
Note:
Remember this figure from last time, shown here is a model of the functional states for these channels. Notice a few states for Na vs two for K.
Conclusions from patch clamp experiments
- Allowed the direct observation of ionic currents flowing through single ion channels
- Both Na⁺ and K⁺ channels are voltage gated
- Thus there must be a voltage sensor in these channels
- Depolarization inactivates Na⁺ channels but not K⁺ channels
Note:
Patch clamp method summary video
Note:
Many genes encode ion channels
- There are hundreds of genes encoding ion channels (e.g. about 100 K⁺ channel genes)
- They have common properties (similarities in amino acid sequence and protein topology)
- They also have variations (differences in ion selectivity, how they are gated, inactivation mechanisms)
Note:
Now everything going on in our nervous systems depends on the function of ion channels. And there are lots of them.
Different ways to gate ion channels
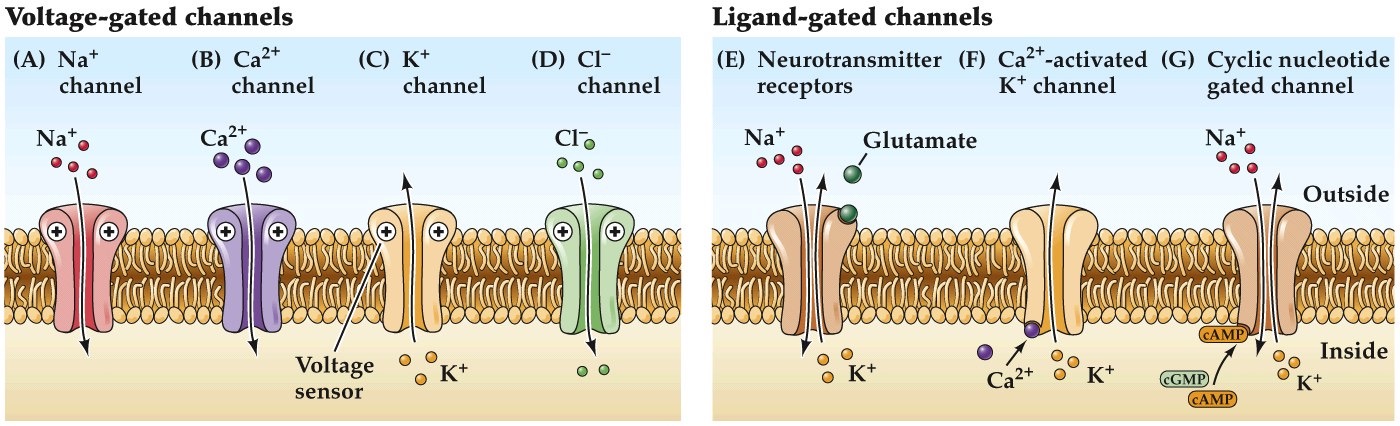
Note:
Different classes of gated ion channels.
voltage gated ion channels, such as we’ve been discussing over the last couple classes.
ligand gated channels such as those that bind neurotransmitters, will talk about more later and next class.
others are ligand gated channels sensitive to chemical signals arising in the cytoplasm of neurons such second messengers like Ca²⁺, cyclic nucleotide cAMP and cGMP.
Lots of variation among ion channels and their properties
- Voltage gated– Na⁺, K⁺, Cl⁻, and Ca²⁺ channels
- Approximately 10 different genes for Na⁺ channels, 16 Ca²⁺, 3–5 Cl⁻ and 100 K⁺ channels
- Different genes may give rise to channels with different properties– e.g. different inactivation times, probability of opening at a given voltages, gating mechanisms
- Can also be multiple splice variants of the same gene
- Creates huge diversity of channels
- How to characterize all these channels?
Note:
Xenopus oocytes
- Large (1 mm in diameter) cell that contains lots of protein synthesis machinery
- Can inject RNA into it and it will express protein encoded by RNA
- Works great for expressing a gene of interest (ion channels!). Can voltage clamp and determine properties of a given channel
- Can make specific mutations in genes and see what happens to function of protein
Note:
If you have a gene for a channel, how do you determine its properties?
frog germ cells
- Need an experimental system where you can express gene of interest functionally and away from other channels
- Xenopus oocytes have been a historical way to do this
Xenopus oocytes for ion channel physiology studies
Inject ion channel mRNA into oocyte ⟶ oocyte makes protein ⟶ patch clamp recordings
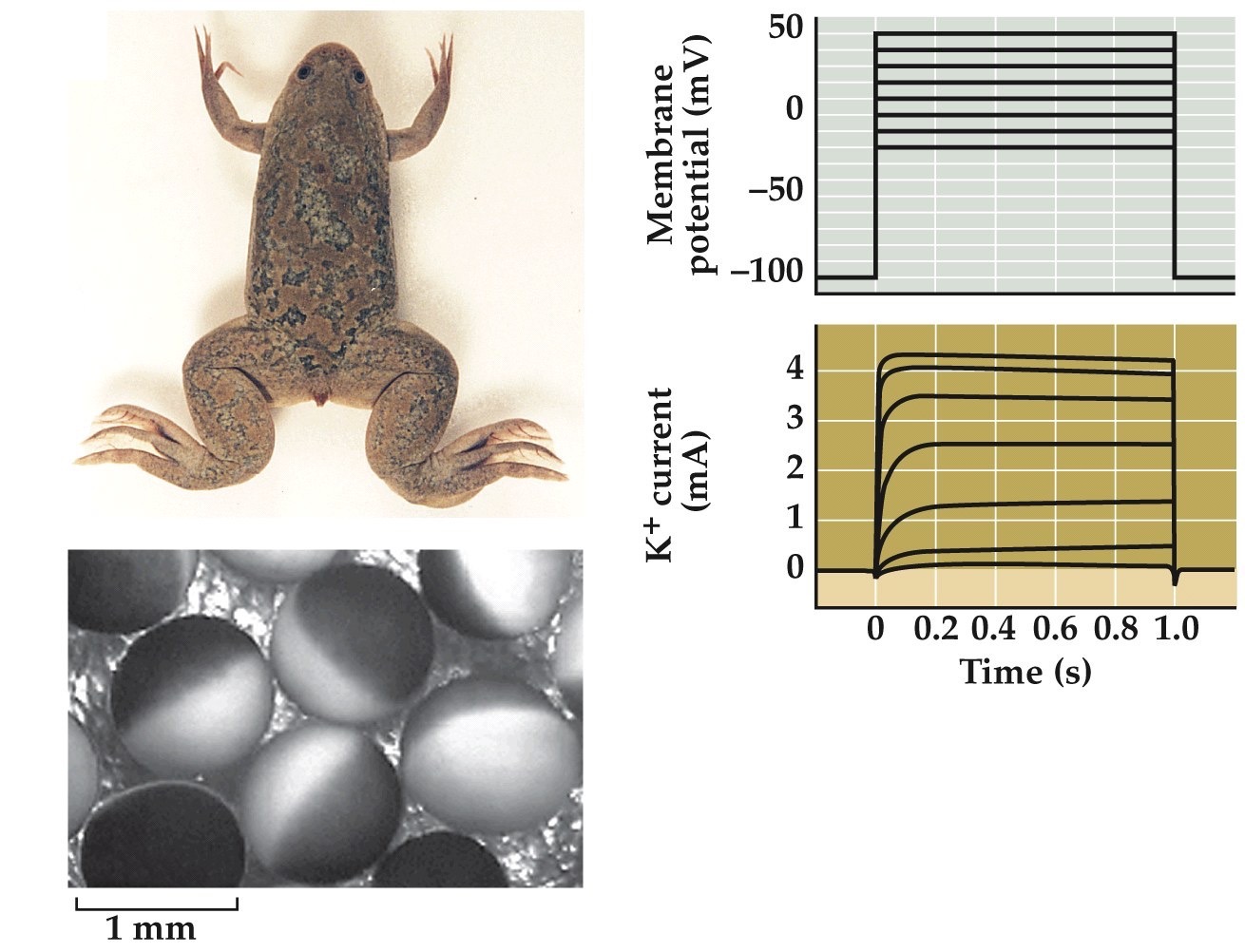
Note:
shows voltage clamp experiment results after expression of a K channel in an oocyte.
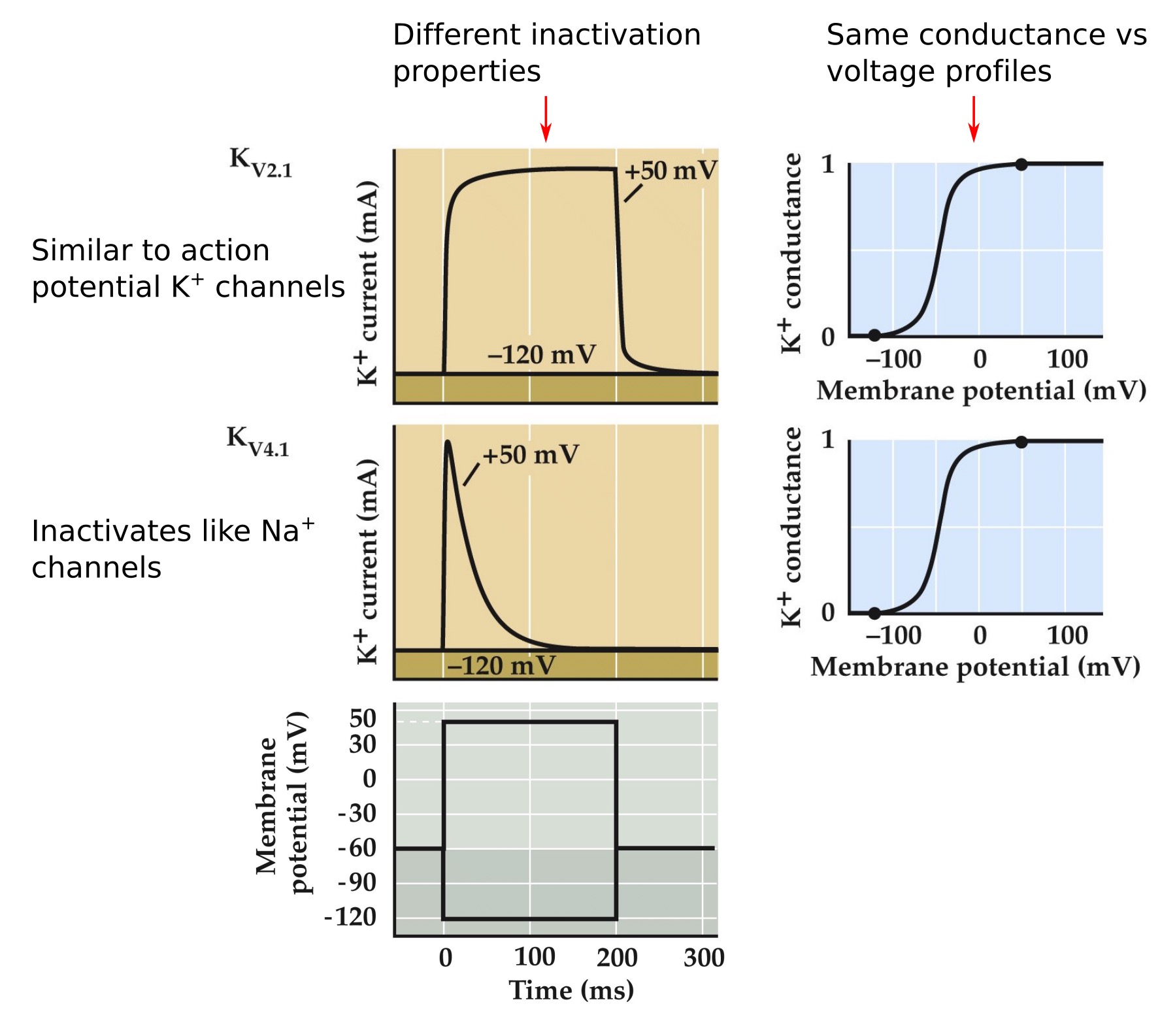
Different K⁺ channels can have diverse properties
Note:
- Kv2.1 show little inactivation and are closely related to the delayed rectifier K channels involved in AP repolarization
from channelpedia:
Kv2.1 is widely expressed in brain and composes the majority of delayed rectifier K+ current in pyramidal neurons in cortex and hippocampus and is also widely expressed in interneurons. Dynamic modulation of Kv2.1 localization and function by a mechanism involving activity dependent Kv2.1 dephosphorylation dramatically impacts intrinsic excitability of neurons.
- Kv4.1 channels inactivate during a depolarization.
a voltage-activated A-type potassium ion channel and is prominent in the repolarization phase of the action potential. This gene is expressed at moderate levels in all tissues analyzed, with lower levels in skeletal muscle.
- inward rectifier K channels allow more K current to flow at hyperpolarized potentials than at depolarized potentials
- human Ether-à-go-go-Related Gene), best known for its contribution to the electrical activity of the heart that coordinates the heart's beating, mediates the repolarizing IKr current in the cardiac action potential).
- HERG channels inactivate so rapidly that current flows only when inactivation is rapidly removed at end of a depolarization
inward rectifier K channels allow more K current to flow at hyperpolarized potentials than at depolarized potentials
Ca activated K channels open in response to intracellular Ca ions
2-P K channels ("two-pore", or KCNK gene family, 50+ genes?) can respond to other signals (e.g. pH changes for the TASK (KCNK3 and KCNK9) channel subtypes) rather than changes in membrane potential and are important in regulating the ongoing membrate potential of neurons at "rest", playing a role in the historically termed "Kleak" current.
https://www.nature.com/articles/35058574
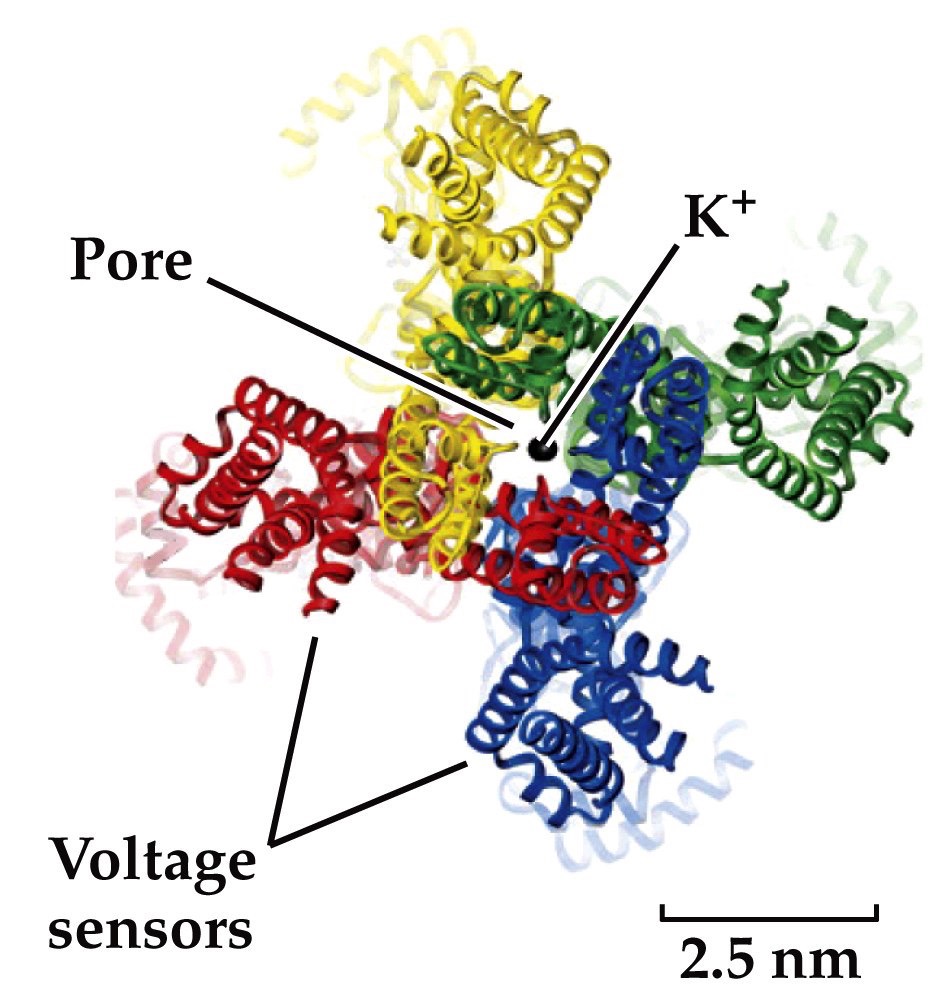
Molecular structures of ion channels
- Multiple membrane spanning domains
- K⁺ channels– 4 subunits come together, (each with 6 transmembrane helices)
- Na⁺ channels– 1 protein with 24 transmembrane helices
- Center has an opening that makes a pore for the ion to flow through
- Contains selectivity filter
- Voltage-gated ion channels also contain a voltage sensitive transmembrane domain
Note:
We’ve learned from biophysical structure studies that in general ion channels have 24 transmembrane peptide domains with…
We can also guess a few characteristics of their structure from the classic voltage clamp and patch clamp studies we’ve discussed over the past couple classes…
- X-ray crystallography
- tool for identifying the atomic and molecular structure of a crystal
- crystalline atoms cause high energy (high frequency/short wavelength) electromagnetic waves (X-rays) to scatter in different directions
- measure intensities and angles of the diffracted beams and compute a 3D model of the electron density in a crystal
- information on mean atomic positions, type of chemical bonds, and more can be extracted
Structure of the bacterial K⁺ channel
- Bacteria have K⁺ channels that are very similar in structure to mammalian K⁺ channels. Main difference is that they are not gated by voltage
- Could be crystallized in the bacterial membrane
- 3D structure tells us a lot about function
- Roderick Mackinnon Nobel Prize in Chemistry 2003
"for structural and mechanistic studies of ion channels"
Note:
prokaryotic
eukaryotic
Structure of the bacterial K⁺ channel
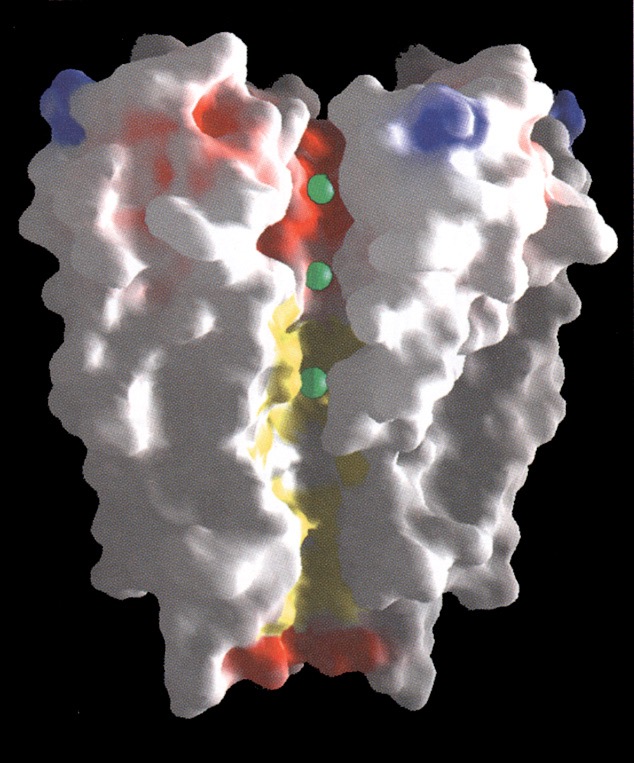
A space-filling model of the KcsA channel, showing the pore. Ions (green balls) tend to occupy three sites in the channel, two in the selectivity filter and one in a pool of water in the center of the channel.
Note:
(Doyle et al, Science 280:69, 1998)
Structure of the bacterial K⁺ channel
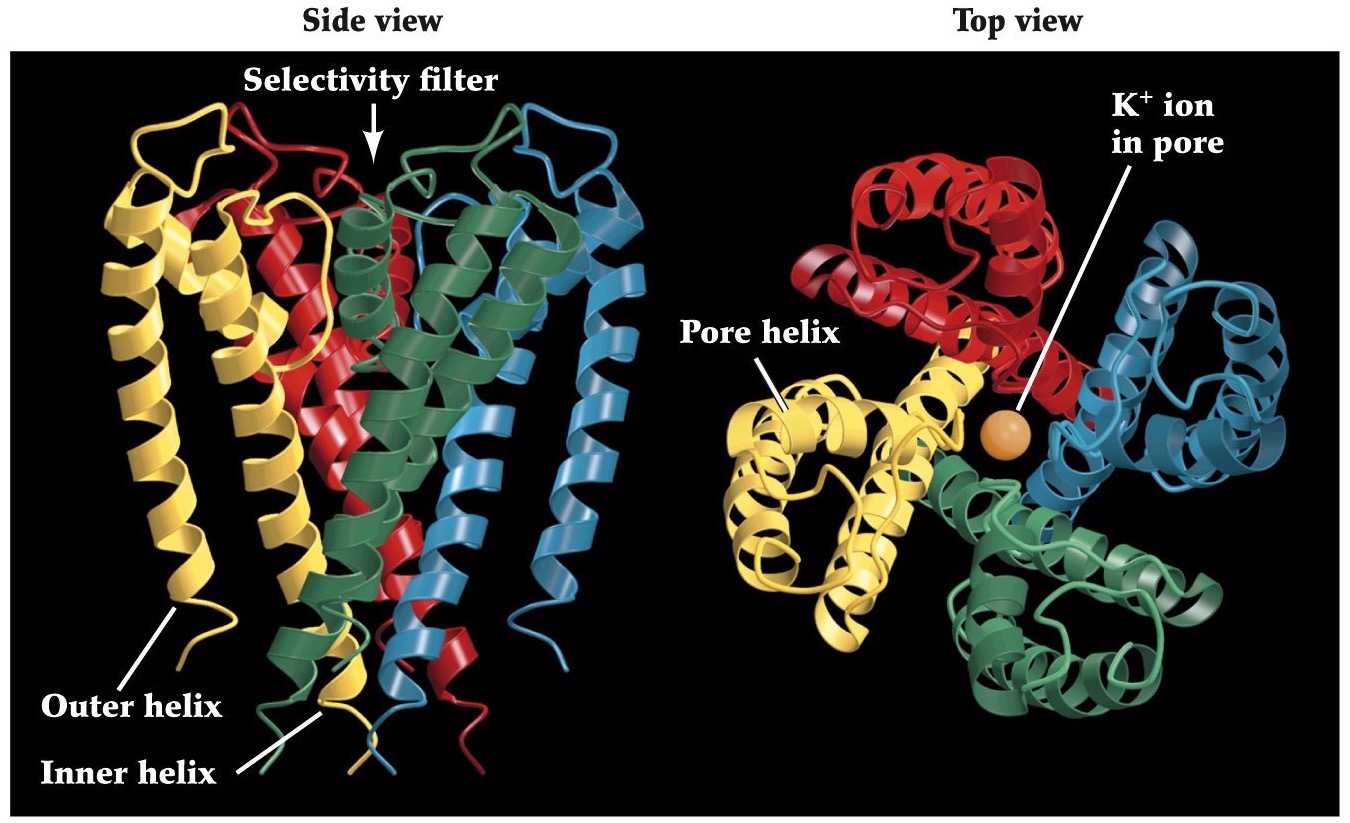
Note:
(Doyle et al, Science 280:69, 1998)
Structure of a bacterial K⁺ channel determined by crystallography
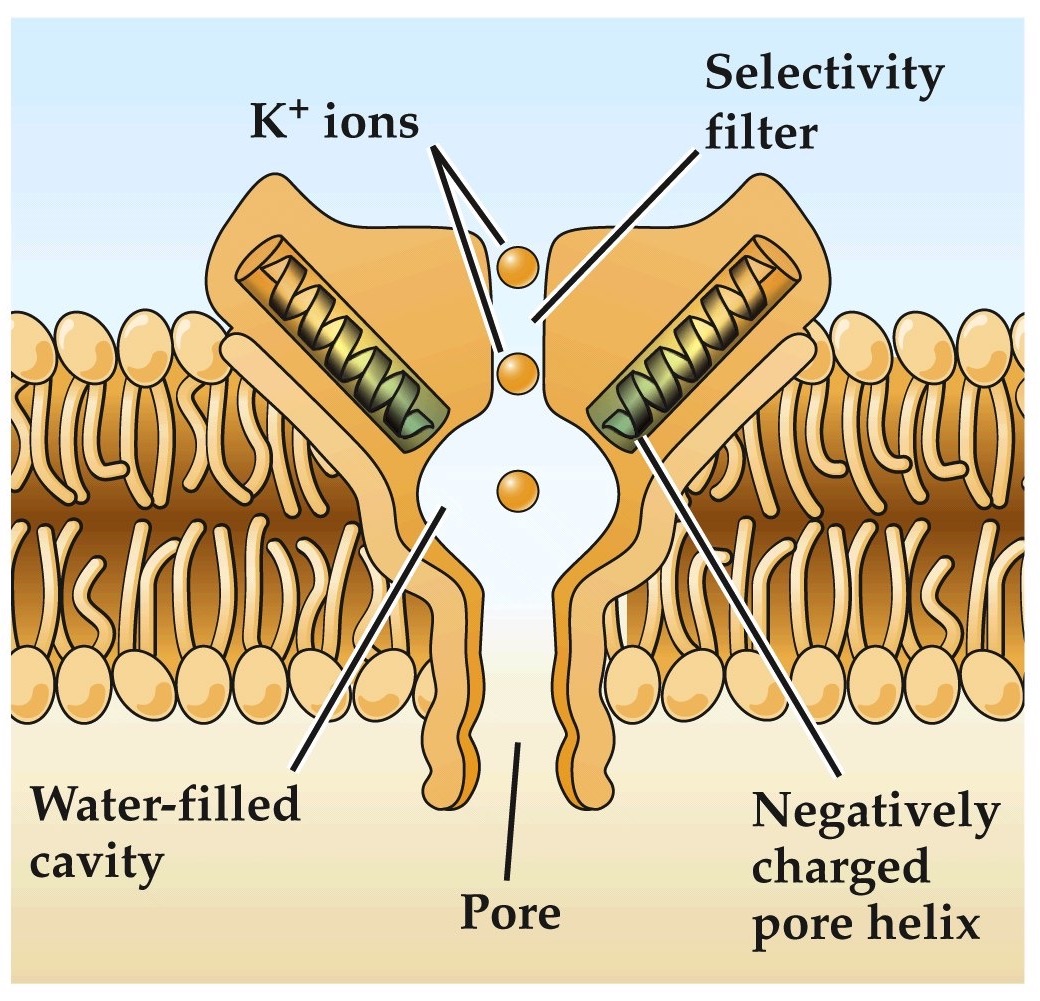
Note:
Simplified model of bacterial K channel, showing you the pore and selectivity filter.
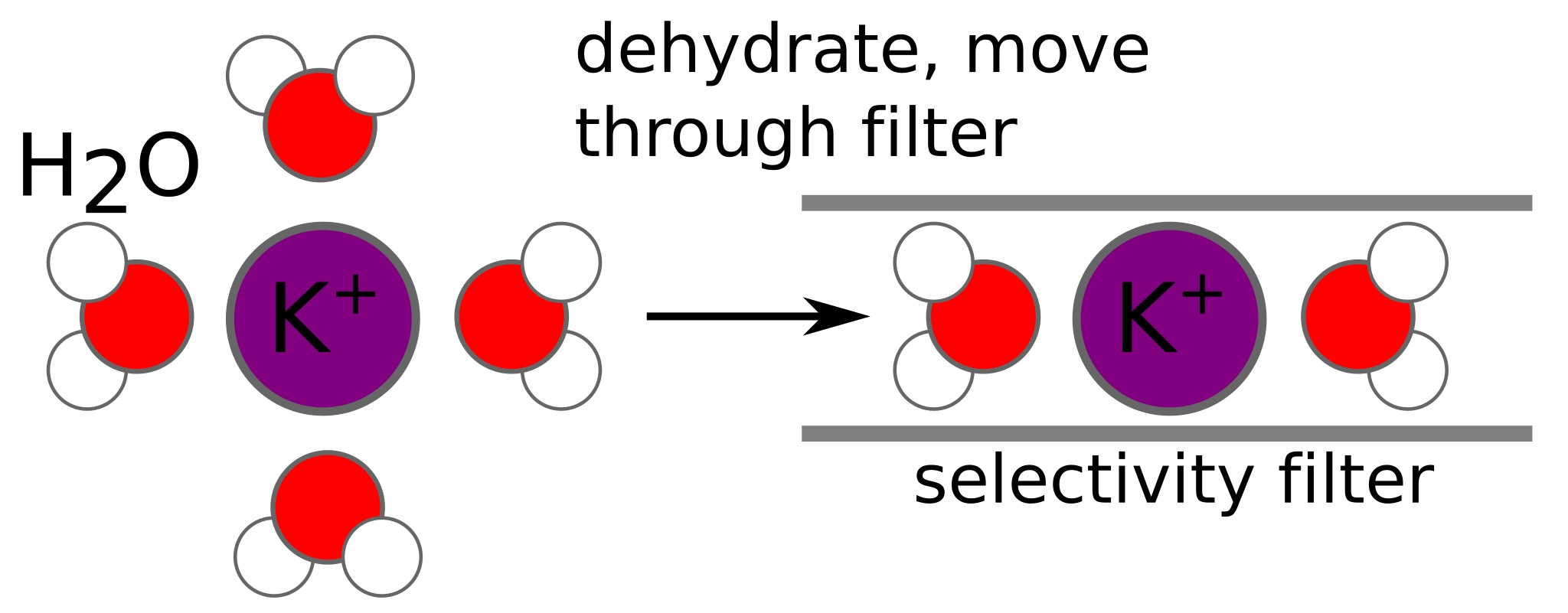
helical domains of channel point negative charges towards cavity allowing K ions to become dehydrated and then push through selectivity filter through electrostatic repulsion.
outside inside helps dehydrate K⁺ ions
Selectivity filter of the K⁺ channel
- Up to 4-6 water molecules form hydration shells around both Na⁺ and K⁺ ions
- Ions move with their hydration shells
- To pass through the potassium channel, an ion must remove most of its surrounding water molecules (dehydrated)
- K⁺ is dehydrated by the K⁺ channel selectivity filter (leaving just two water molecules– one at front and one at back)
- Na⁺ has a more stable water shell, binding H2O more strongly and thus has a larger effective diameter— would require more dehydration energy than K channel pore region can provide
| ion | ion diameter (nm) free | ion diameter hydrated |
|---|---|---|
| Na | 0.19 | 0.52 |
| K | 0.27 | 0.46 |
JA, CC0
Note:
remember water is a polar molecule. Has a net dipole moment of opposing charges in the hydrogen-oxygen bonds.
Larger cations cannot traverse the pore region, smaller cations like Na cannot enter the pore because the walls are just too far apart to stabilize a dehydrated Na ion long enough to pass through.
Na is the most hydrated ion with 4 to 6 water molecules in the first shell. Binds water strongly, making a stable hydration shell and moving together with the cation. Any sodium movement is followed by H2O movement (water retention, excretion).
Potassium ion is larger, having 8 more electrons shielding positively charged nucleus, thus K⁺ makes transient associations with water rather than a discrete hydration layer. Helps explain higher permeability across cell membrane for K⁺.
ion | ion diameter (nm) free | ion diameter hydrated
--- | ---------------------- | -------------------
Na | 0.19 | 0.52
K | 0.27 | 0.46
a quote from http://web-books.com/MoBio/Memory/Channel.htm:
To pass through the potassium channel, an ion must remove most of its surrounding water molecules, leaving only two - one at the front and another at the back.
The selectivity filter of the sodium channel is slightly larger than that of the potassium channel. It may accommodate a Na⁺ ion attached with three water molecules, but not enough for a K⁺ ion attached with three water molecules.
one more quote from http://web-books.com/MoBio/Memory/Channel.htm:
In the sodium channel, the Na⁺ ion is more permeable than the K⁺ ion. This is because the selectivity filter of the sodium channel is slightly larger than that of the potassium channel. It is large enough to accommodate a Na⁺ ion attached with three water molecules, but not enough for a K⁺ ion attached with three water molecules. Therefore, to pass through the sodium channel, the Na⁺ ion needs to remove only three, but the K⁺ ion has to remove four, water molecules from its first hydration shell. The required dehydration energy for the K⁺ ion is greater than the Na⁺ ion. In calcium channels, the permeability of monovalent cations (Na⁺ and K⁺) is about three orders of magnitude smaller than the Ca²⁺ permeability. This ion selectivity does not seem to involve hydration, because Ca²⁺ is more heavily hydrated than Na⁺, and the unhydrated diameters of Ca²⁺ and Na⁺ are almost identical. Then, how could calcium channels select Ca²⁺ over Na⁺? Although the permeability of monovalent cations in the calcium channel is quite small at normal ionic concentrations, large monovalent cationic current can be observed in the absence of Ca²⁺ and other divalent cations. This suggests that the calcium channel is basically permeable to both divalent and monovalent cations, but the selectivity arises from competition between ions. The calcium channel may contain a negatively charged binding site to facilitate ion conduction. The monovalent cations simply cannot compete with Ca²⁺ for this binding site. This idea has been confirmed experimentally. In the calcium channel, if a negatively charged glutamate residue in the pore-lining region is mutated into a positively charged lysine, the calcium channel becomes more permeable to Na⁺ than Ba2+
This explains the selectivity but not the voltage sensor
Atomic masses
1H < 2He
3Li < Be < B < 6C < N < 8O < 9F < 10Ne
11Na < 12Mg < Al < 14Si < P < S < 17Cl < 18Ar
19K < 20Ca
37Rb < 38Sr
55Cs < 56Ba
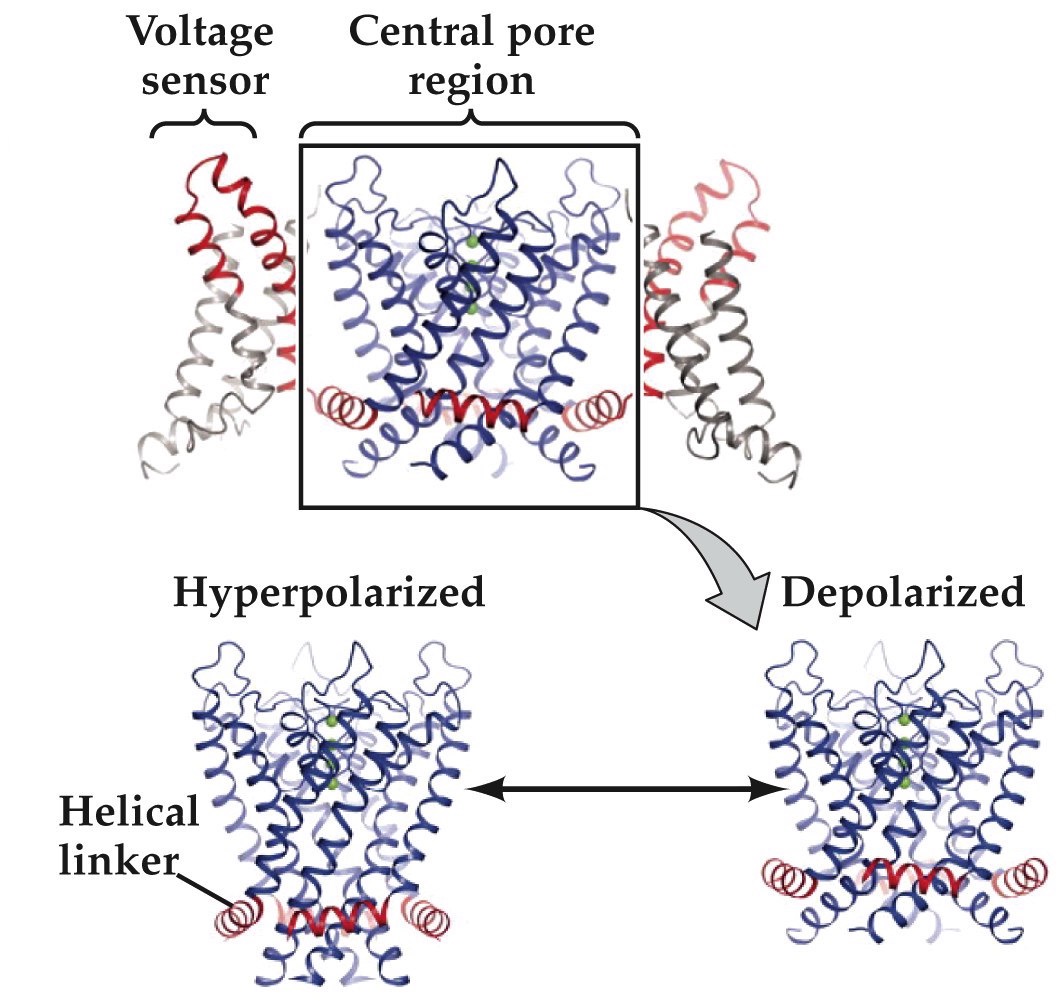
Structure of a mammalian voltage-gated K⁺ channel
Note:
Now we know from what we’ve learned over the past couple classes that unlike bacteria, neurons have K⁺ channels that are gated by voltage
http://web-books.com/MoBio/Memory/Channel.htm :
There are many types of potassium channels. The one involved in the generation of action potentials is composed of four subunits, each is homologous to the Shaker protein (Fig. 3.2). The hydrophobicity profile indicates that it contains six hydrophobic segments, designated as S1 - S6. These segments are likely to be the transmembrane domains. Other experimental results suggests that the P-region is lining the channel pore.
Structure of a mammalian voltage-gated K⁺ channel
Note:
Molecular structures of ion channel proteins
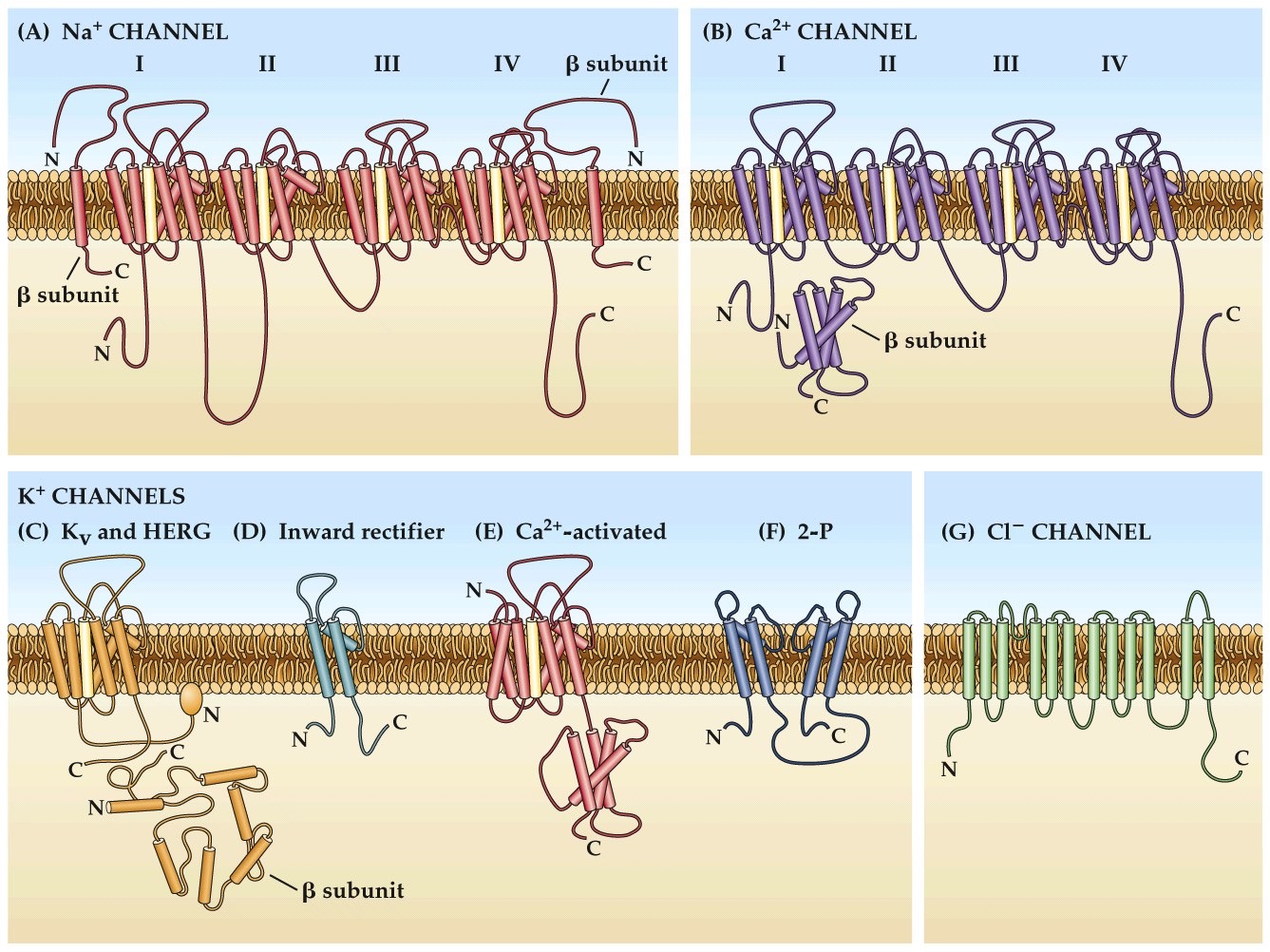
Note:
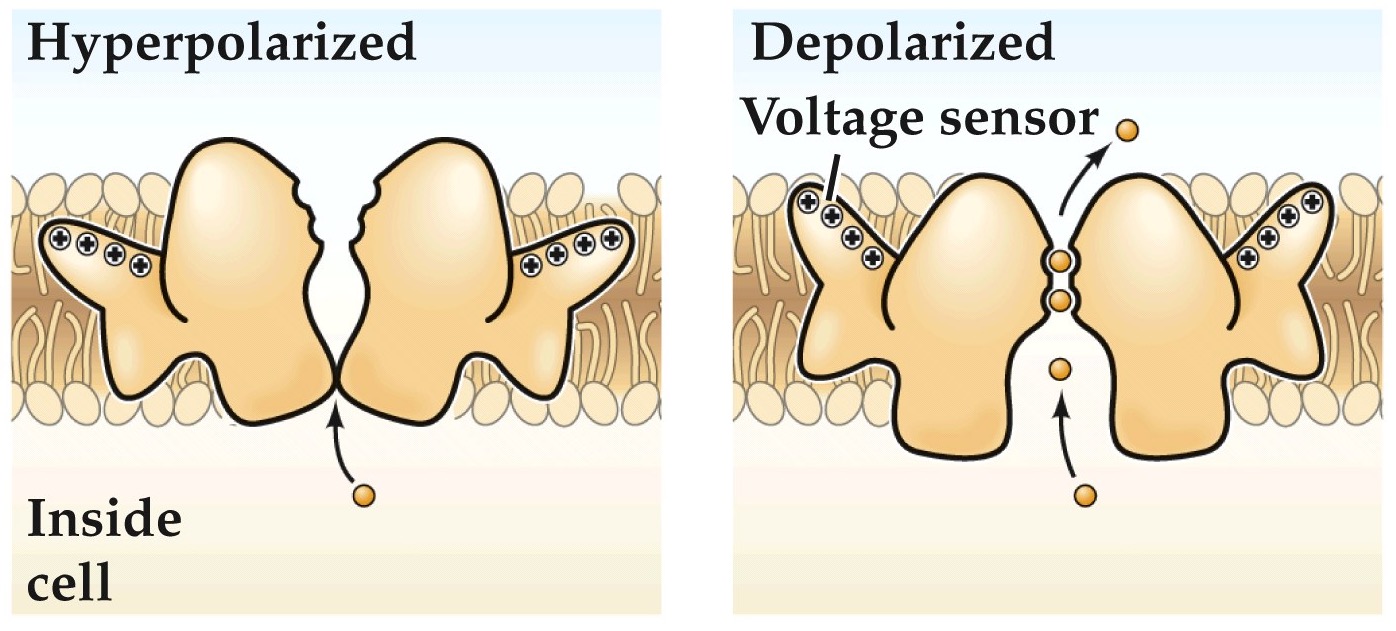
Yellow are voltage sensing tm domains
4–8 positively-charged amino acids in the S4 domain. Experiences force in a transmembrane electric field. Is the electric-field sensor for voltage-dependent gating.
K channels are diverse
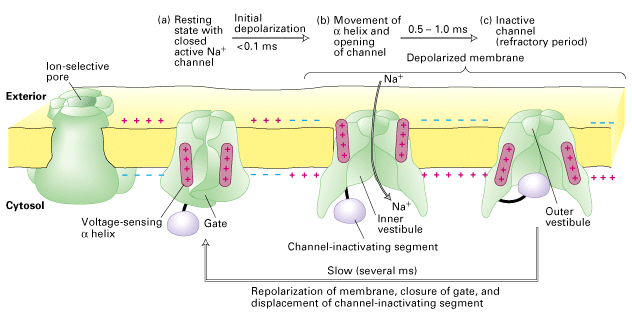
How do Na⁺ channels inactivate?
- Contains an activation gate that binds to the channel in the intracellular region and blocks the channel
- Activation gate changes conformation (closes/swings shut) to block channel only during the channel’s open state
- Therefore, at resting Vm channel is closed and activation gate is open
- After depolarization, the channel opens and Na⁺ ions go through. After a little bit of time (~ 1 ms) the activation gate swings shut to block channel
http://www.nature.com/nature/journal/v475/n7356/full/nature10238.html
Note:
The theory is that the inactivation gate “swings” shut, turning off the channel
The physical structure of voltage gated Na channels has only recently begun to be solved, with the results so far fitting the models for Na channel opening and inactivation.
Sodium channel inactivation cycle
Note:
Figure 21-13 Lodish 4th edition OR Figure 7-33 Lodish 5th edition. Structure and function of the voltage-gated Na⁺ channel.
Toxins that poison ion channels
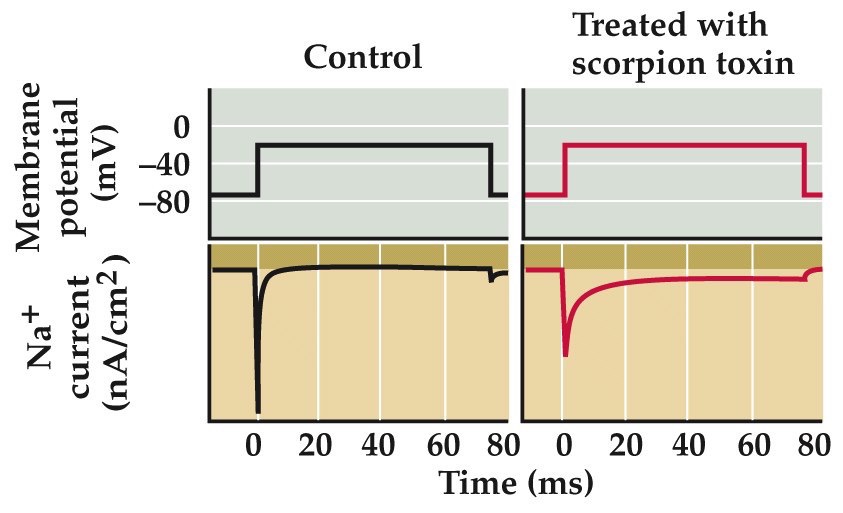
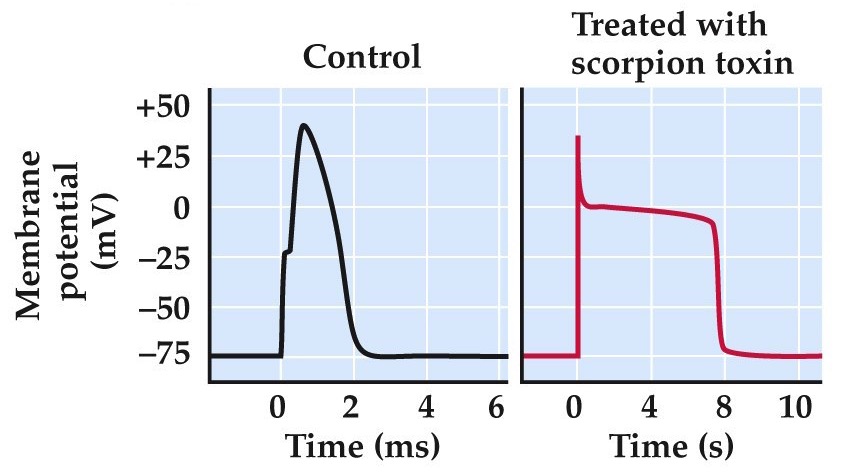
http://www.nature.com/news/rodent-immune-to-scorpion-venom-1.14014
Note:
already learned about tetrodotoxin from puffer fish. blocks voltage gated Na channels underlying the AP
saxitoxin similar (homologue) to ttx, produced by dinoflagellates and possible effects from ‘red tide’ or eating shellfish that have injested these dinoflagellates.
scorpions paralyse prey by injecting alpha-toxins (left panels). Slow inactivation of Na channels, prolonging the AP and messing up information flow in CNS. Beta-toxins in scorpion venom shift the voltage dependence of Na channel activation (right panel), causing Na channels to open at potential much more negative than normal inducing uncontrolled AP firing.
Some alkaloid toxins (batrachotoxin, produced by S. American frogs) do both of these mechanisms.
Similar toxins from plants (aconitine from buttercups, veratridine from lilies) and insecticidal toxins (pyrethrins) produced by chrysanthemums and rhododendrons.
dendrotoxin from wasps affects K channels
apamin from bees K channels
charybdotoxin from scorpions K channels
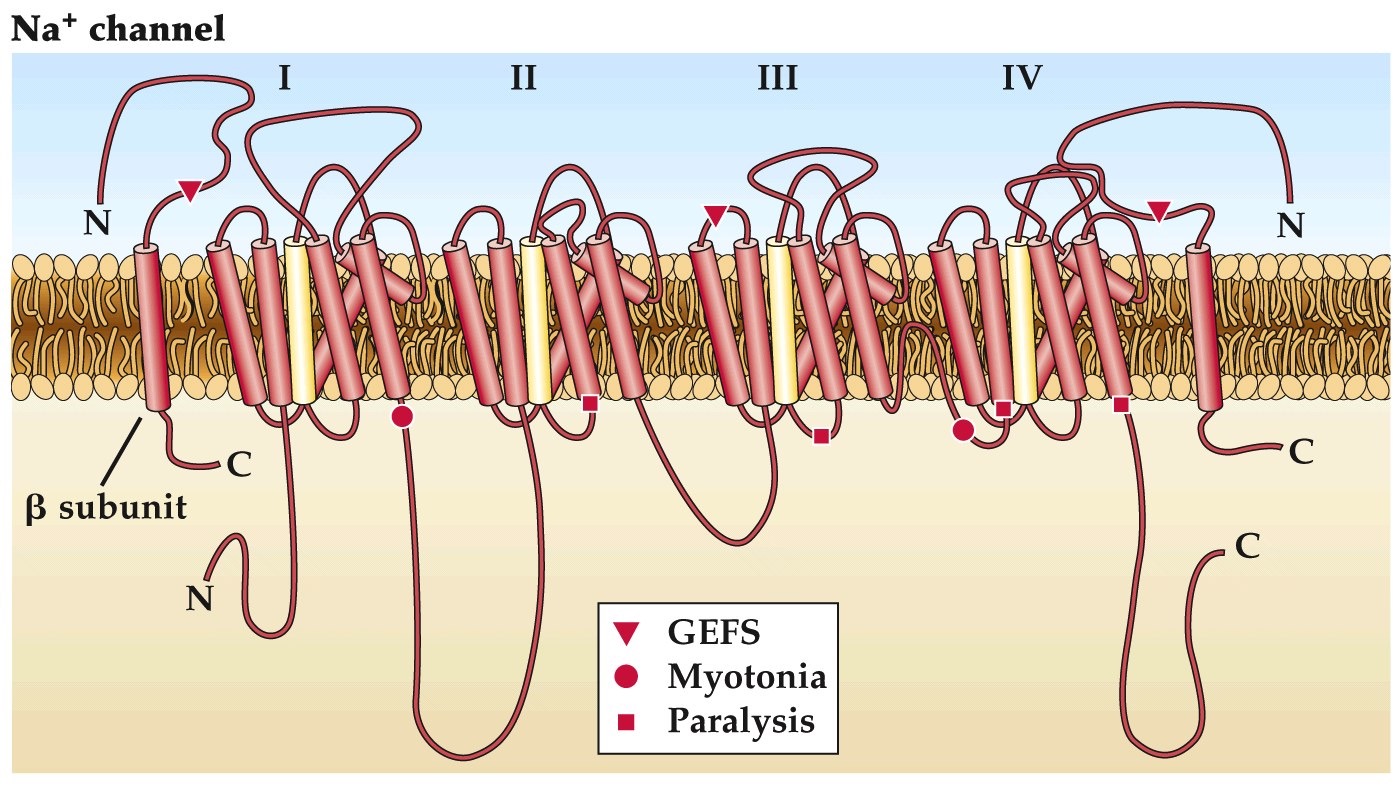
Diseases caused by ion channel mutations
- Channelopathies: genetic diseases resulting from mutations in ion channel genes
- e.g. >50 neurological disorders, >40 cardiac disorders (Kim 2014)
GEFS: generalized epilepsy with febrile seizures, begins at infancy and continues through puberty. Mapped to two mutations, one on an alpha Na channel subunit and one on a beta subunit. Cause slowing of sodium channel inactivation
Myotonia: muscle contractions
Paralysis: muscle weakness
Note:
More than 20 different inhereited diseases from from mutations in Na channels alone. Cystic fibrosis results from chloride channel dysfunction (and altered fluid movements, chloride gradients often used for cell volume, fluid movements).
ataxia: greek for ‘without order’ or ‘incoordination’. Movement coordination problems.
paralysis: muscle weakness
myotonia: muscle contraction
--
Epilepsy can result from mutated Na⁺ channels
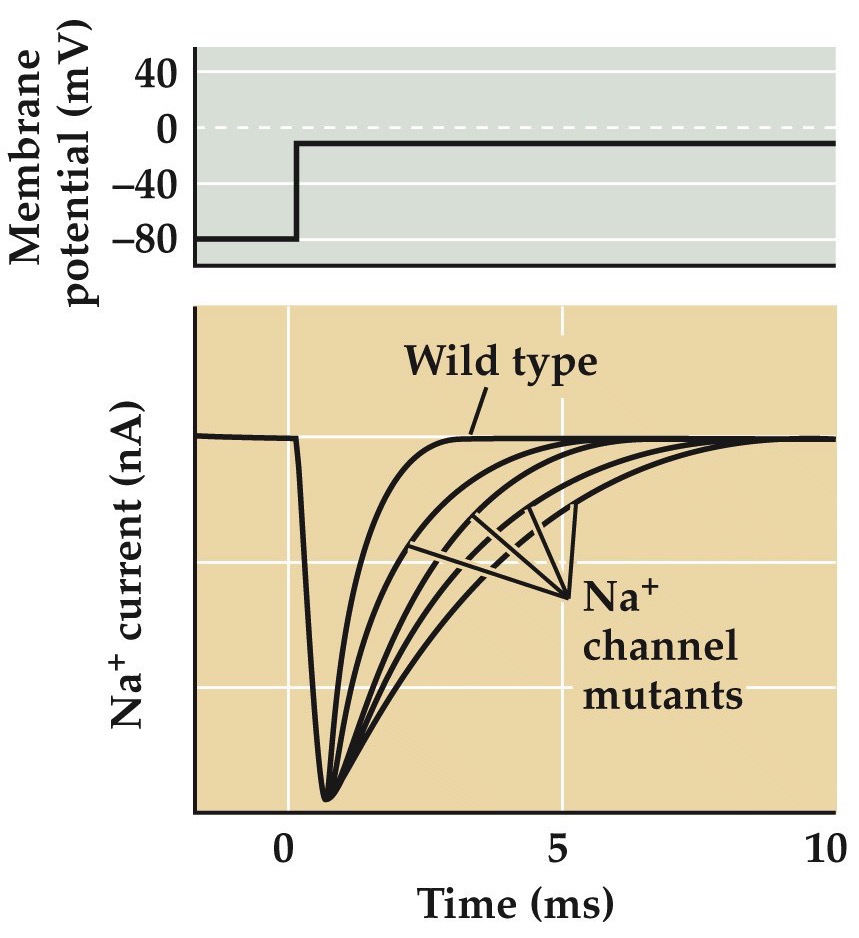
Note:
You can see the the slower inactivation kinetics in this figure here in patch clamp recordings from normal and a number of different Na channel mutants. This slowing of Na inactivation is just enough to mess up spike patterns in single neurons and elicit hyperexcitability that results in seizures in networks of connected neurons.
GEFS: generalized epilepsy with febrile seizures
Ion transporters
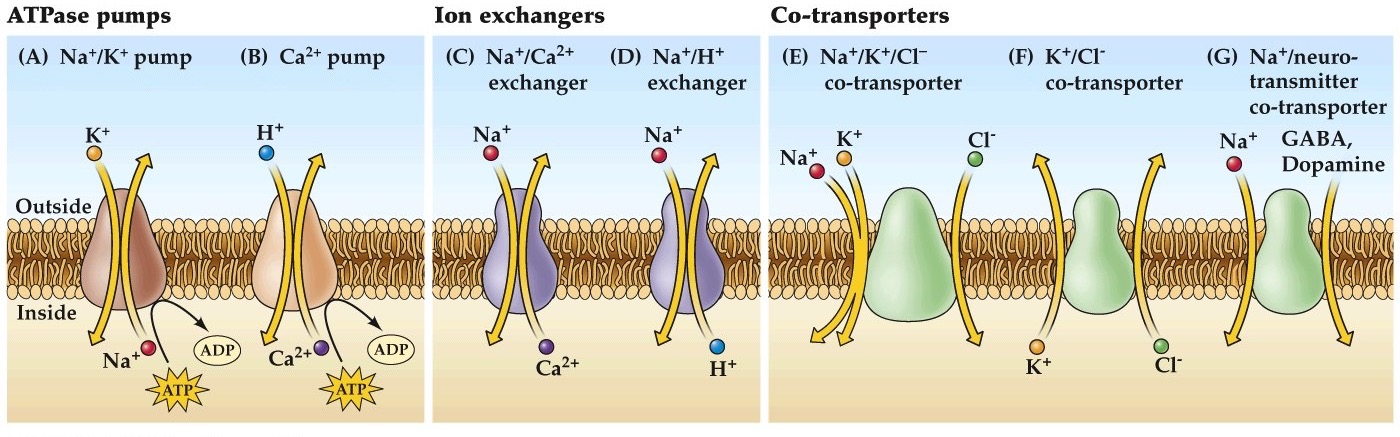
Note:
Lastly let's remind ourselves of the importance of ion transporters in maintaining the concentration gradients across the nerve cell membrane. We've previously discussed the active transporter the Na/K pump that is crucial for maintaining Na/K gradients but there are others that maintain gradients for other physiologically relevant ions like Cl, Ca.
Remember these transporters are all very slow compared to ion channels, requiring several milliseconds to move a few ions compared to thousands of ions per second conducted across the membrane for an ion channel.
Crystal structure for Na/K channel with either K bound in the central pore or Na was just solved in 2009 and 2013 respectively (Shinoda et al, Nature 2009) Nyblom et al. Science 2013)
Ouabain, plant 'arrow' poison traditionally from africa from the Acokanthera schimperi and Strophanthus gratus plants. Binds to the Na+/K+ pump. Cardiac dysfunction ensues.
Used together with
Radioactive Na efflux measurements and radioactive K influx measurements used with ATP synthesis inhibitors (e.g dinitrophenol) to help demonstrate that an active Na/K pump is responsible for producing ion conentraiton gradients in squid axon (Hodgkin and Keynes 1955).