32 KiB
Neurotransmitters and receptors
- Neurotransmitters, ligands for receptors; >100 molecules
- small molecule transmitters
- acetylcholine (ACh), amino acids, biogenic amines, purines
- neuropeptides
- various polypeptides, 3–36 amino acids in length
- small molecule transmitters
- Neurotransmitter receptors
- ionotropic, 'ligand-gated' ion channels
- metabotropic, G-protein coupled receptors that modulate separate ion channels
Note:
Diving a bit deeper into the structure and function of neurotransmitter (NT) receptors now...
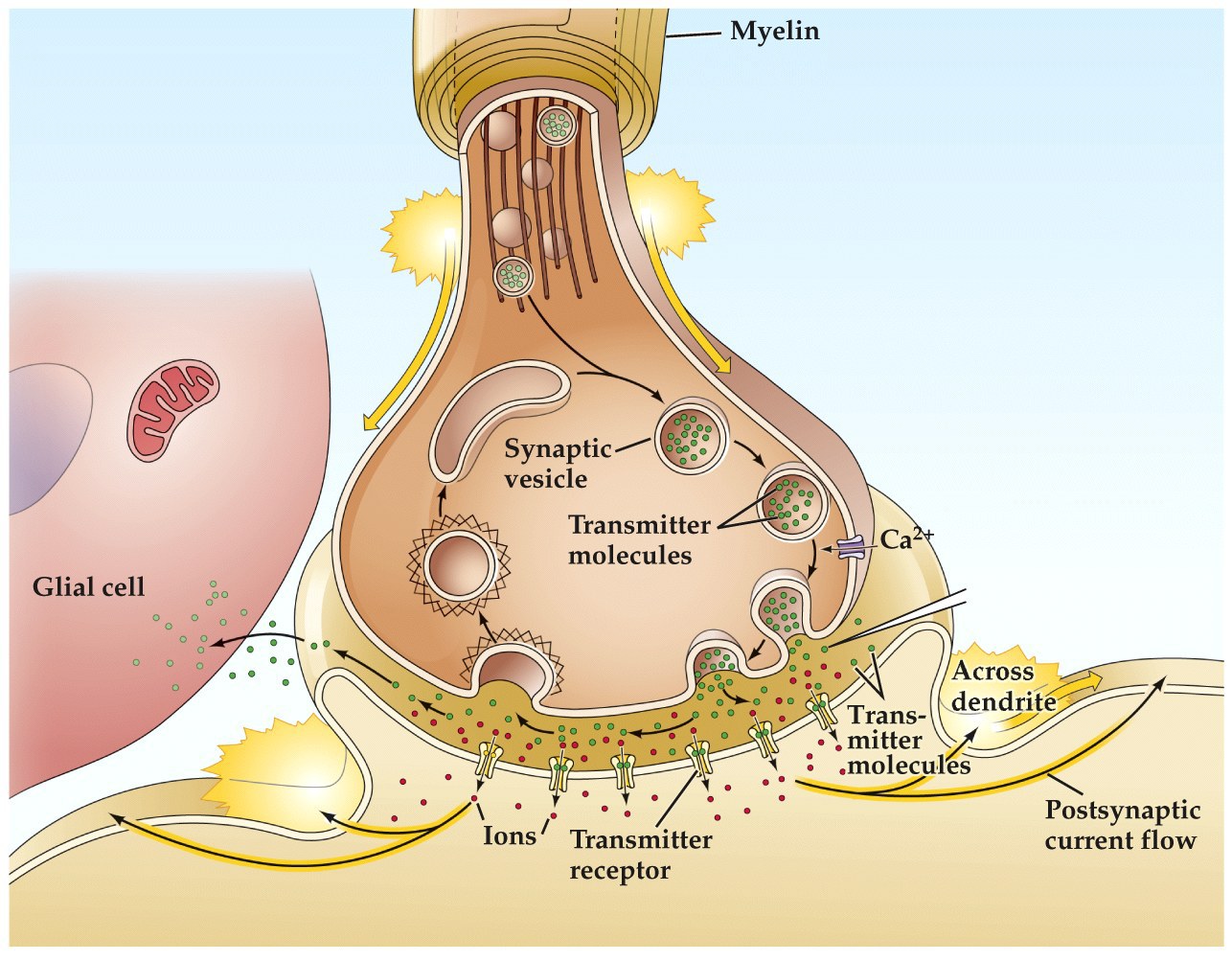
For synaptic transmission, NT receps are generally located in the post-synaptic membrane (though there are exceptions, e.g. some transmitter receptors may be located on pre-synaptic membrane or at non synaptic site in the cell).
Two classes of NT receptors.
In either case, NT binding will result in ion channels opening and ion flux across the post-synaptic membrane. Whether this results in hyperpolarization or depolarization of the membrane will be due to the types of ions flowing through the channels and their respective electrical/chemical driving forces (Nernst)
Changing the postsynaptic membrane potential inturn affects the electrochemical driving forces regulating ion flux. So currents may change amplitude and direction during the course of a postsynaptic potential. Read on...
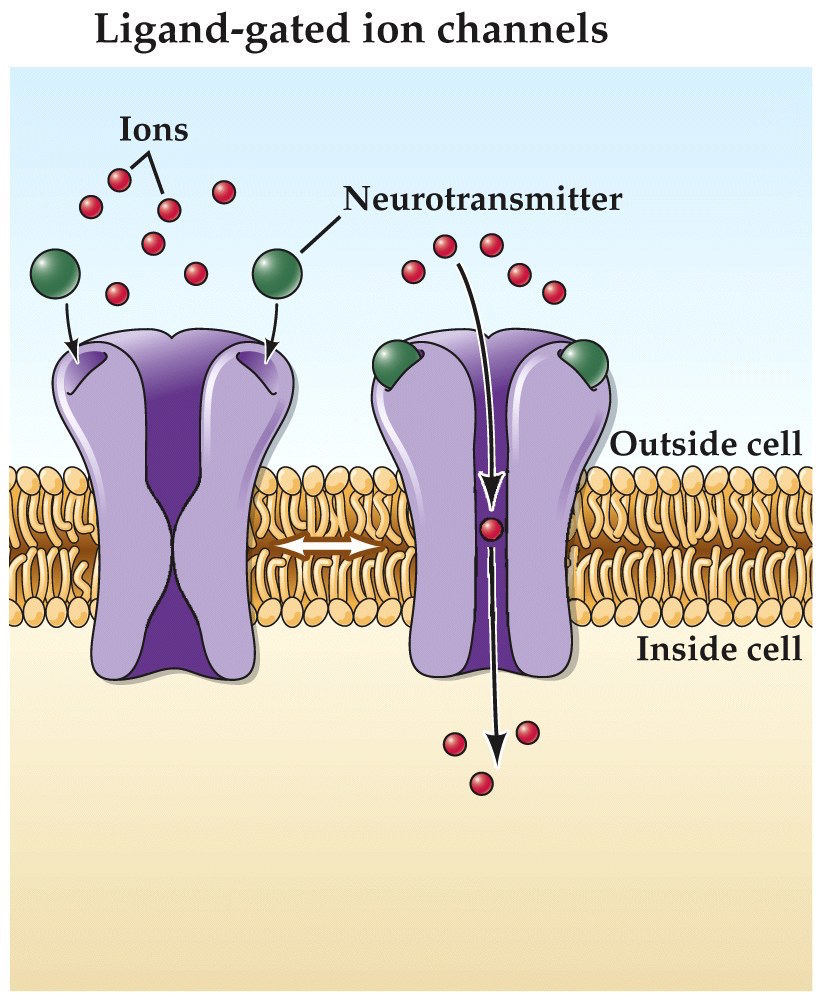
Ionotropic neurotransmitter receptors
- Neurotransmitter binds receptor
- Channel opens, allowing ions to flow through
Note:
The ionotropic receptors are the ones you’ve probably seen in our synaptic diagrams so far, where NT binds directly to an ion channel pore, causing it to open and allow ions to move through the pore.
- neurotransmitter binds
- channel opens
- ions flow across membrane
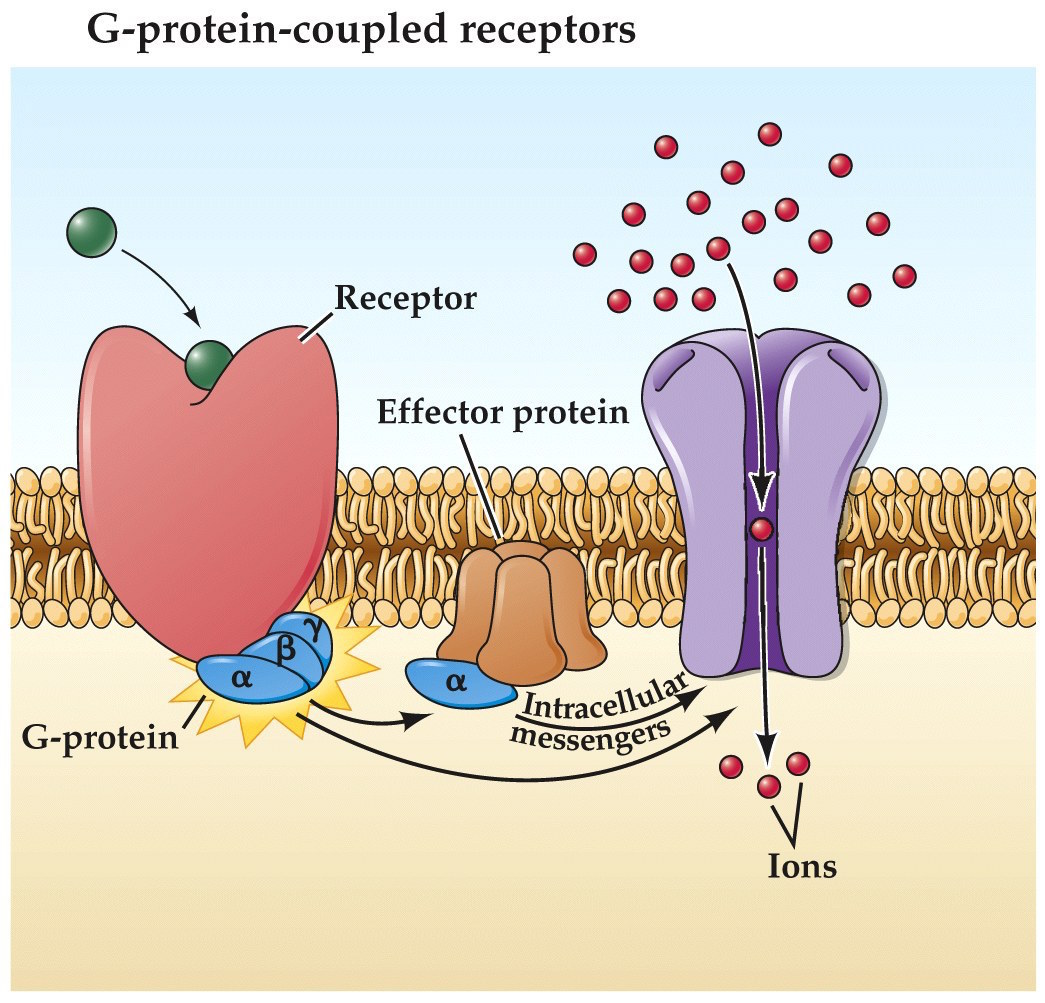
Metabotropic neurotransmitter receptors
- G-protein coupled receptor signalling results in modulation of nearby ion channels for metabotropic receptors.
Note:
The photoelectric response (from bacteria to human retinal photoreceptors) depends on metabotropic modulation of ion channel function. Also the response to odorants in nasal epithelium for olfaction as well as certain chemosensory receptors for taste. And the receptors for neuropeptides, as well as biogenic amines like dopamine, norepinephrine, histamine, and serotonin
Metabotropic transmitter receptors are G-protein coupled receptors, also known as seven-transmembrane domain receptors in your cell biology courses.
- neurotransmitter binds
- g protein binds and is activated
- g protein subunits or intracellular messengers modulate ion channels
- ion channel opens
- ions flow across membrane
Effector enzymes for activated G-proteins include:
-
Gs: --> adenylyl cyclase --> ATP--> cAMP --> PKA => incr prot phosphor
-
Gq: phopholipase C --> [DAG --> PKC; IP3 --> Ca^2+^] => incr prot phosphor, Ca binding proteins
-
Gi: --< adenylyl cyclase --< cAMP --< PKA => decr prot phosphor
-
Or guanylyl cyclase (GTP->cGMP) --> Protein kinase G etc.
-
All G-protein receptor activations lead to downstream second messsaging (cAMP, diacylglycerol, IP3) --> protein kinases, Ca2+ --> leading to phosphorylation state changes including... ion channels
-
Three amplification steps here! (receptor production of G proteins, adenylyl cyclase production of cAMP, protein kinase substrate phosphorylation). Source signal amplification.
-
3% of our genome is codes for protein phosphorylation state genes (500 protein kinases and 200 protein phosphatases)
- cAMP dependent protein kinases (PKA)
- Ca^2+^ - calmodulin dependent protein kinase type II (CaMKII predominant in neurons, most abundant protein component of the post synaptic density)
- Protein kinase C (PKC)- activated by Ca^2+ (moves PKC from cytosol to membrane) and diacylglycerol (DAG) and then phosphorylates substrates
--
Neurotransmitter receptors video summary
Note:
Nicotinic acetylcholine receptors (nAChR)
- Ionotropic receptor
- Acetylcholine (ACh) binds the nAChR– this opens the channel
- ACh causes nAChR to open transiently and stochastically (patch clamp studies)
- An action potential causes lots of ACh molecules to be released simultaneously, transiently opening many nACh receptors
- The summed current flow into the muscle cell is called the end plate current (EPC). Current flow changes the transmembrane potential of the muscle, the end plate potential (EPP), which triggers an action potential
Note:
So to understand the properties of ionotropic neurotransmitter receptors lets start with the nicotinic ACh receptor (abbreviated nAChR).
nACh Receptors are ionotropic or ligand-gated receptors where the ligand is ACh and are the receptor you’ve heard the most thus far, being the one that underlies end plate currents at the neuromuscular junction that cause end plate potentials in muscle cells.
- stochastic
- having a random probability distribution or pattern that may be analyzed statistically but may not be predicted precisely
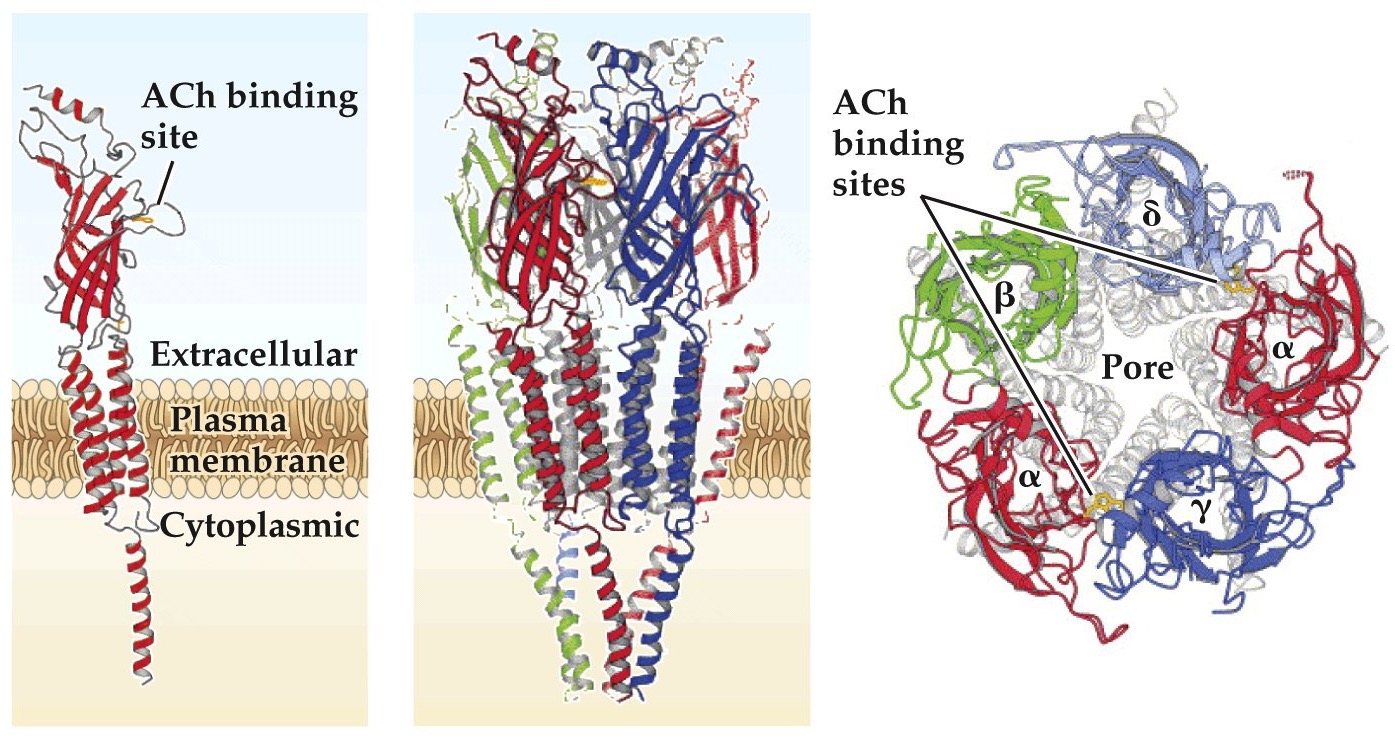
nAChR
- Pentamer- 5 subunits to make a pore. Selective for cations
- Pore diameter 10x greater than voltage-gated Na⁺ channels (3 nm vs 0.3 nm)
- Nicotine mimics ACh to stimulate receptor, an agonist
- nicotinic ACh receptors (nAChR) produce excitatory postsynaptic potentials (EPSPs or EPPs)
- Many toxins specifically bind and block nAChR; these are antagonists
- alpha-bungarotoxin (snake venom)– binds to alpha subunit of nAChR very tightly and prevents ACh from activating it
Note:
As we’ve shown in our examples earlier the nAChR receptor is a non-selective cation channel. Or another way to think of it is that it is selective for cations.
5 subunits
nAChR permeable to Na+, K+, and Ca2+
In physiological solution, calcium flux estimated to be 2% of total current through nAchR. For comparison calcium flux is estimated to be 7% of the current in the voltage gated L-type calcium ion channel. But with high density clustering of many nAchRs at muscle end plate synapses, total calcium flux through these channels could influence the local environment significantly https://doi.org/10.1523/JNEUROSCI.10-10-03413.1990
This Ca^2+^ permeability depends on subunit composition of the nAchR pentamer. mammalian α9α10 receptors receptors show higher calcium ion selectivity (important function in cochlear hair cells) https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4245820/
from [#Picciotto:2000]:
some subtypes of nAChR in the brain (those containing the b2 subunit) are located diffusely throughout the membrane of the neuron, with no obvious concentration at the synaptic junction (Hill et al. 1993).
a number of alpha and beta subunits have expression throughout brain (medulla, superior colliculus, cortex, beta2 subunit expression 'very high' in thalamus). Only alpha3 KO mice have high mortality [#Picciotto:2000].
[#Picciotto:2000]: Picciotto, M. R., Caldarone, B. J., King, S. L., and Zachariou, V. (2000). Nicotinic receptors in the brain. Links between molecular biology and behavior, Neuropsychopharmacology, 22(5), 451-65. PMID 10731620
Low (nM) concentrations of nicotine are found in the blood of moderate smokers (Henningfield et al. 1983). These are sufficient to enhance excitatory transmission in cultures of neurons from the medial habenula or the hippocampus (Gray et al. 1996; McGehee et al. 1995) [#Picciotto:2000]
Many effects of nicotine probably through presynaptic or preterminal nAChRs instead of through postsynaptic AChRs (Léna et al. 1993; Marshall et al. 1997; McGe- hee et al. 1995; Summers and Giacobini 1995; Vidal and Changeux 1993; Wonnacott et al. 1990; Yang et al. 1996) [#Picciotto:2000]
Most effects of nicotine go through nAChR
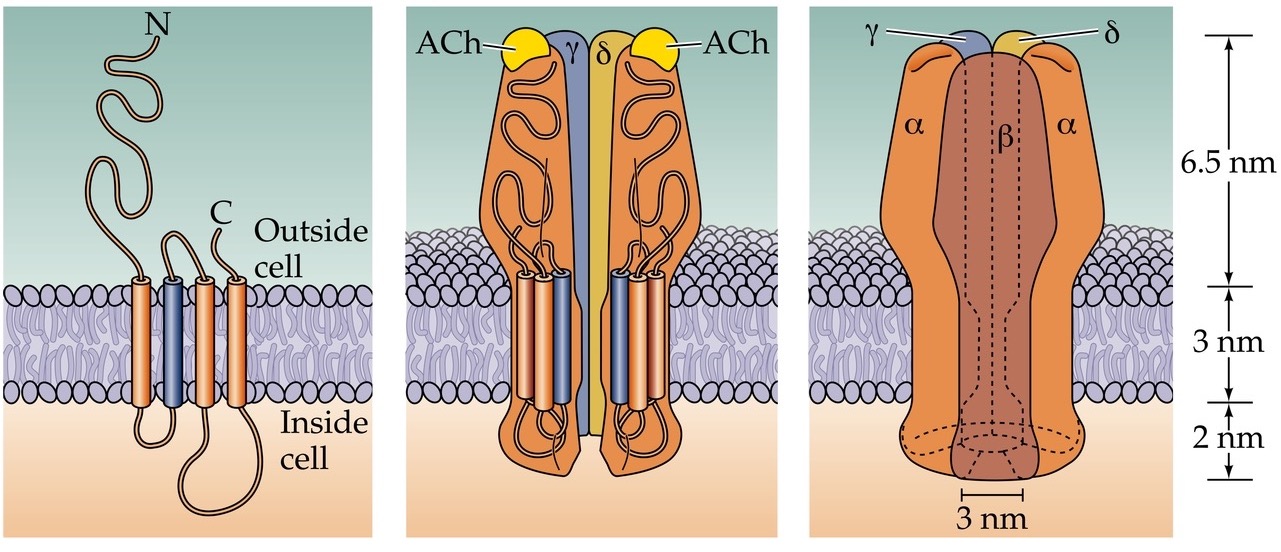
Structure of the nACh receptor
- 5 subunits come together to make a pore
- Each subunit has 3-4 membrane spanning domains
- In muscles the receptor has 2α, β, δ, γ, ε subunits. The α subunits bind ACh, both need to be bound for channel to open. α subunits also binds bungarotoxin and nicotine
- Multiple isoforms for each subunit, depending on which isoform is in channel get different properties
- In neurons its slightly different. 5 subunits 3α:2β. Bungarotoxin only inhibits muscle nACh receptors
Note:
The alpha subunits bind ACh.
Muscle nAChR
- Pentamers of 2α1, β1, γ, δ in fetal mammals vs. 2α1, β1, δ, ε in adult mammal
- ACh, nicotine, curare, and bungarotoxin binding sites are on the α1 subunits
Changes in subunit composition during development.
curare is a competitive antagonist.
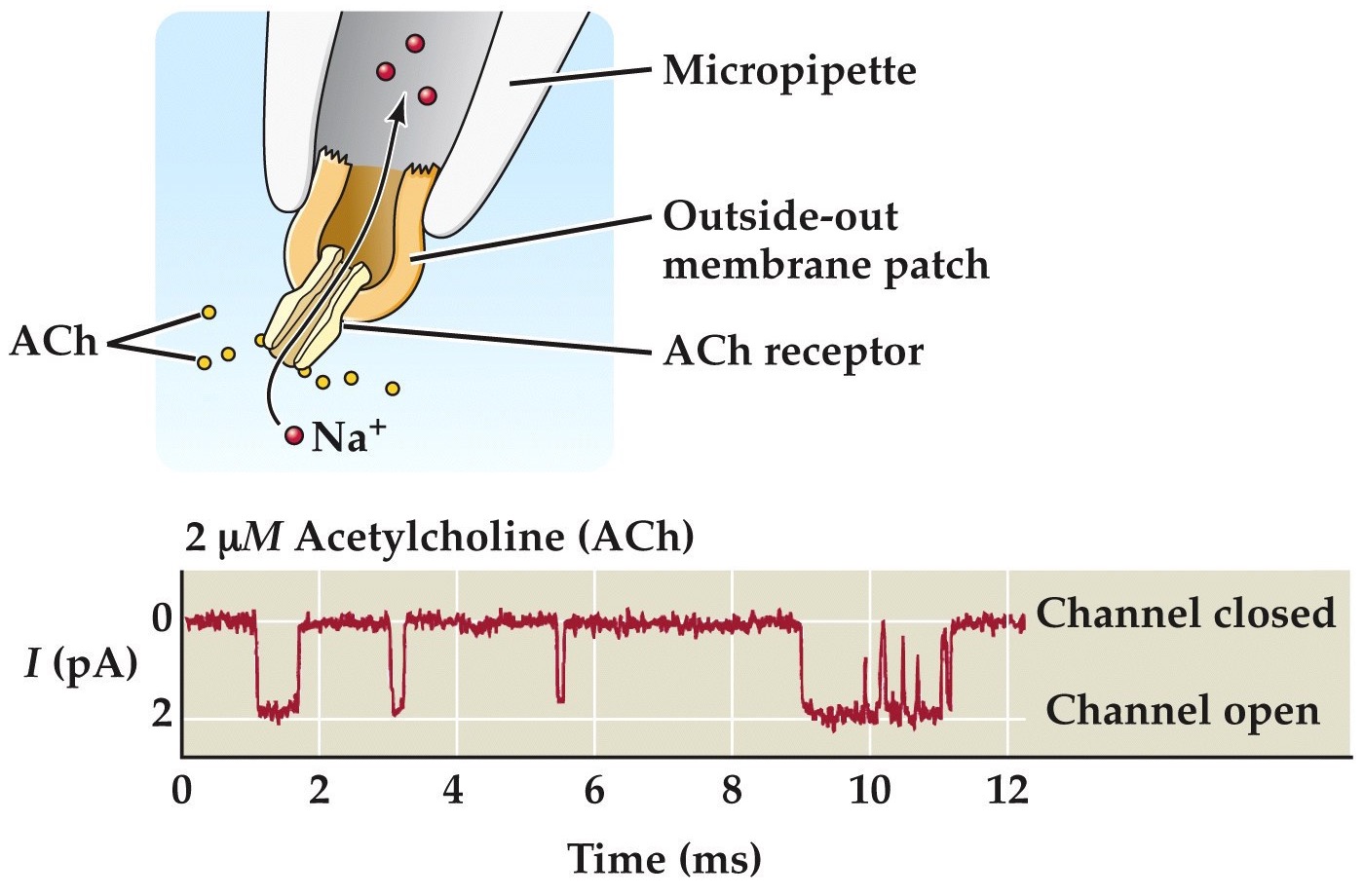
Patch clamping shows ACh gated currents through nicotinic ACh receptors
Note:
The binding of a neurotransmitter to its receptor usually opens (sometimes closes) ion channels.
The figure shows a simple case. In the absence of ACh, the nAChR is closed. In the presence of high [ACh] (the channel always has ACh bound), the channel opens and closes. These repeated brief openings are seen as downward deflections corresponding to inward current. Notice the current amplitudes in this patch clamp trace below are unitary or quantal indicating that a single channel is being recorded in this case...
These look like microscopic currents you get in single channel patch clamp recordings like we discussed previously.
If this piece of membrane and channel is from a muscle cell than a bunch of these currents put together are the ones that give rise to the end plate potentials we for muscle cells before.
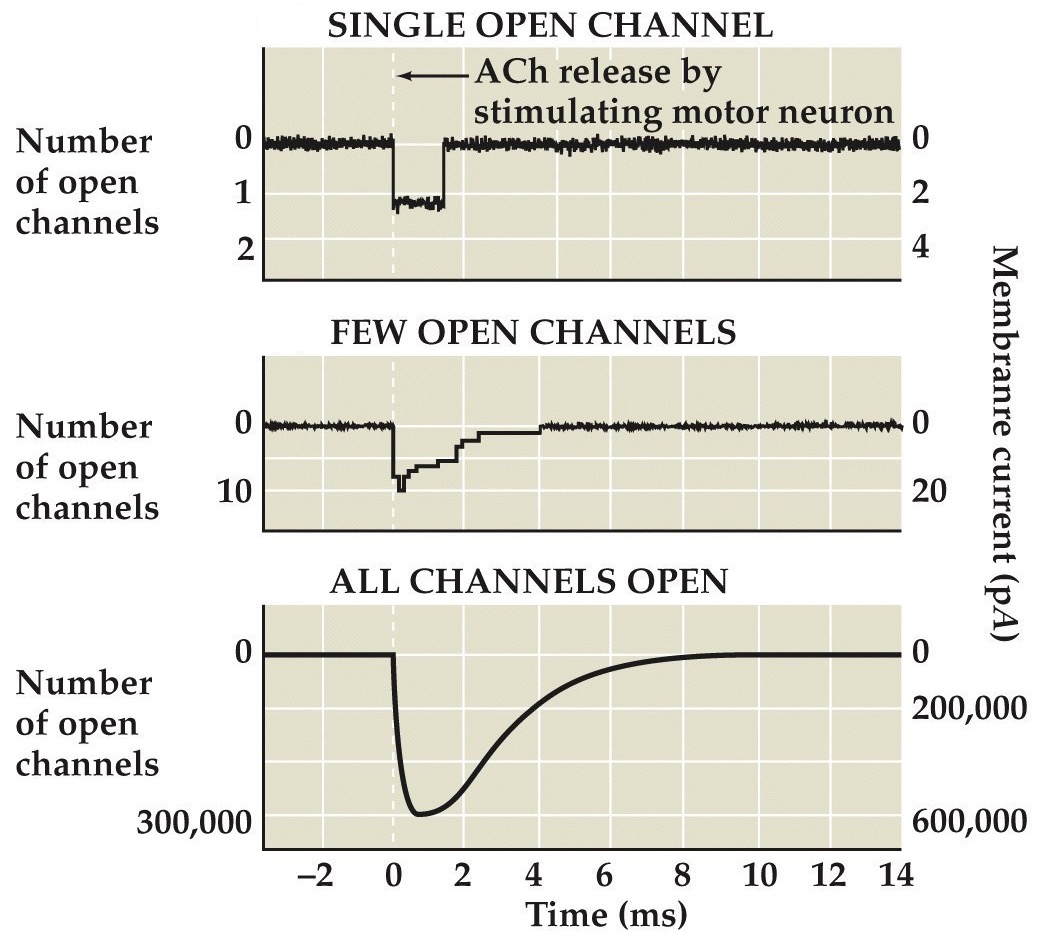
Activation of nAChR at neuromuscular synapses
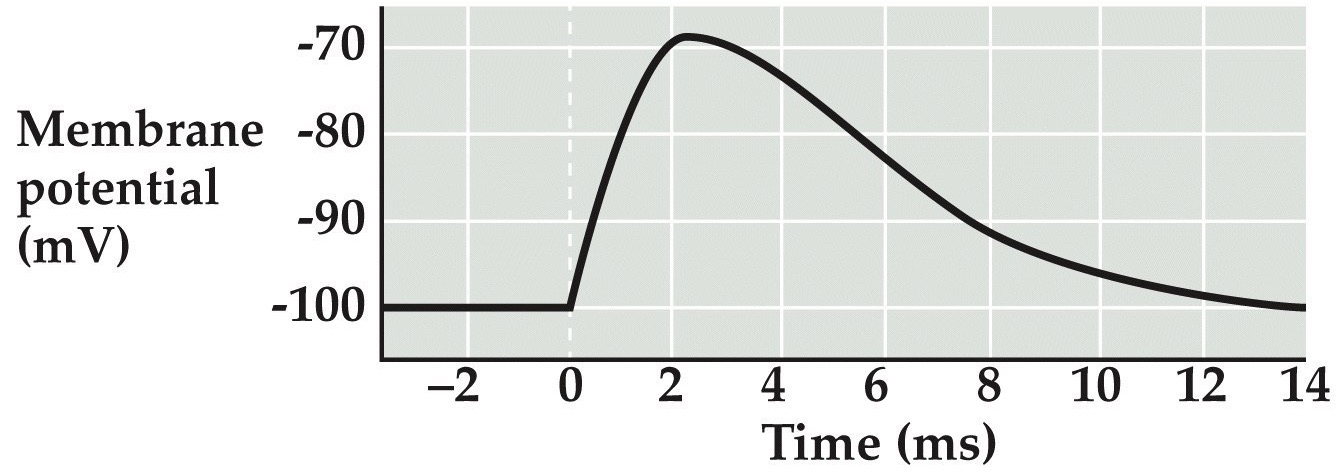
Note:
Imagine we are doing an experiment where we stimulate a motor neuron and we record end plate currents in a muscle cell...
...then the traces on the left show inward currents through these ionotropic ACh channels in the muscle cell, showing the currents stemming from a single channel, 10 channels, and hundreds of thousands of channels. Notice the amplitudes of the currents scale.
...and the panel on the right shows postsynaptic potential change or end plate potential produced by the EPC as we discussed previously
As we will learn shortly, the channel opened by ACh lets mostly Na⁺ through resulting in these inward currents that depolarize the muscle cell, resulting in EPPs and typically resulting in APs as we’ve discussed before.
from http://www.ncbi.nlm.nih.gov/books/NBK21586/:
Two factors greatly assisted in the characterization of the nicotinic acetylcholine receptor. First, this receptor can be rather easily purified from the electric organs of electric eels and electric rays; these organs are derived from stacks of muscle cells (minus the contractile proteins) and thus are richly endowed with this receptor. (In contrast, this receptor constitutes a minute fraction of the total membrane protein in most nerve and muscle tissues.) Second, α-bungarotoxin, a neurotoxin present in snake venom, binds specifically and irreversibly to nicotinic acetylcholine receptors.
- acetylcholine causes opening of a cation channel in the receptor capable of transmitting 15,000 – 30,000 Na⁺ or K⁺ ions a millisecond
What ions flow through the nicotinic ACh receptor?
- Nernst equation– the equilibrium potential of a cell for ion x is the potential at which the electrochemical driving forces is balanced for ion x (i.e there is no net flow of ion x at the equilibrium potential Ex)
- Thus if one measured the ACh dependent current flow at different potentials, one could determine the membrane potential (Vm) where there is no net ion flux (Ix = 0). This is called the reversal potential or Erev
- The end plate current (EPC) at the muscle cell must therefore be IACh and is equal to the driving force on an ion multiplied by its permeability (remember Ohm's law: I = gV)
- IACh = gACh(Vm – Erev)
- Predicts that current will be inward at potentials more negative than Erev, becomes small at potentials approaching Erev, and then becomes outward at potentials more positive then Erev
Note:
Now using our good friend the Nernst eqn, which you can recall is…
Since we know there isn’t any net flow of an ion x, at the Ex, we can measure the ACh dependent currents at different potentials and figure out the potentials at which current flow is 0.
When we are talking about the potential at which postsynaptic currents like the endplate current reverses from inward net ion flux to outward net ion flux, we call this potential the reversal potential denoted Erev.
We can call the endplate current then the IAch or the current flowing through the ACh receptor at skeletal muscle endplate membrane and IAch is therefore equal to the driving force (which is the difference between Vm and Erev) multiplied by the permeability for ACh gAch.
This would then predict that current will be inward at potentials more negative than Erev…
- Predicts that current will be negative (inward) at potentials more negative than Erev, becomes small at potentials approaching Erev, and becomes positive (outward) at potentials more positive then Erev.
Measure postsynaptic (end plate) currents while stimulating motor neuron
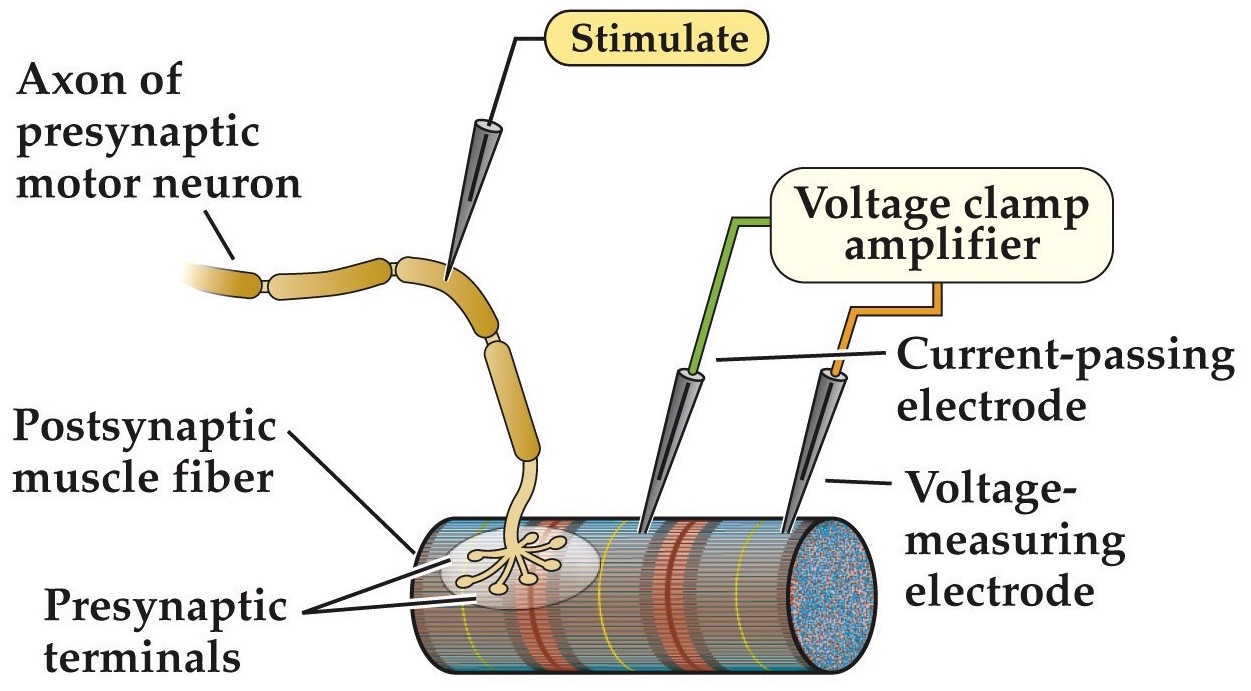
Note:
A postsynaptic muscle fiber is voltage clamped to control the muscle fiber’s membrane potential, while the presynaptic neuron is stimulated to cause ACh release at the end plate synapse.
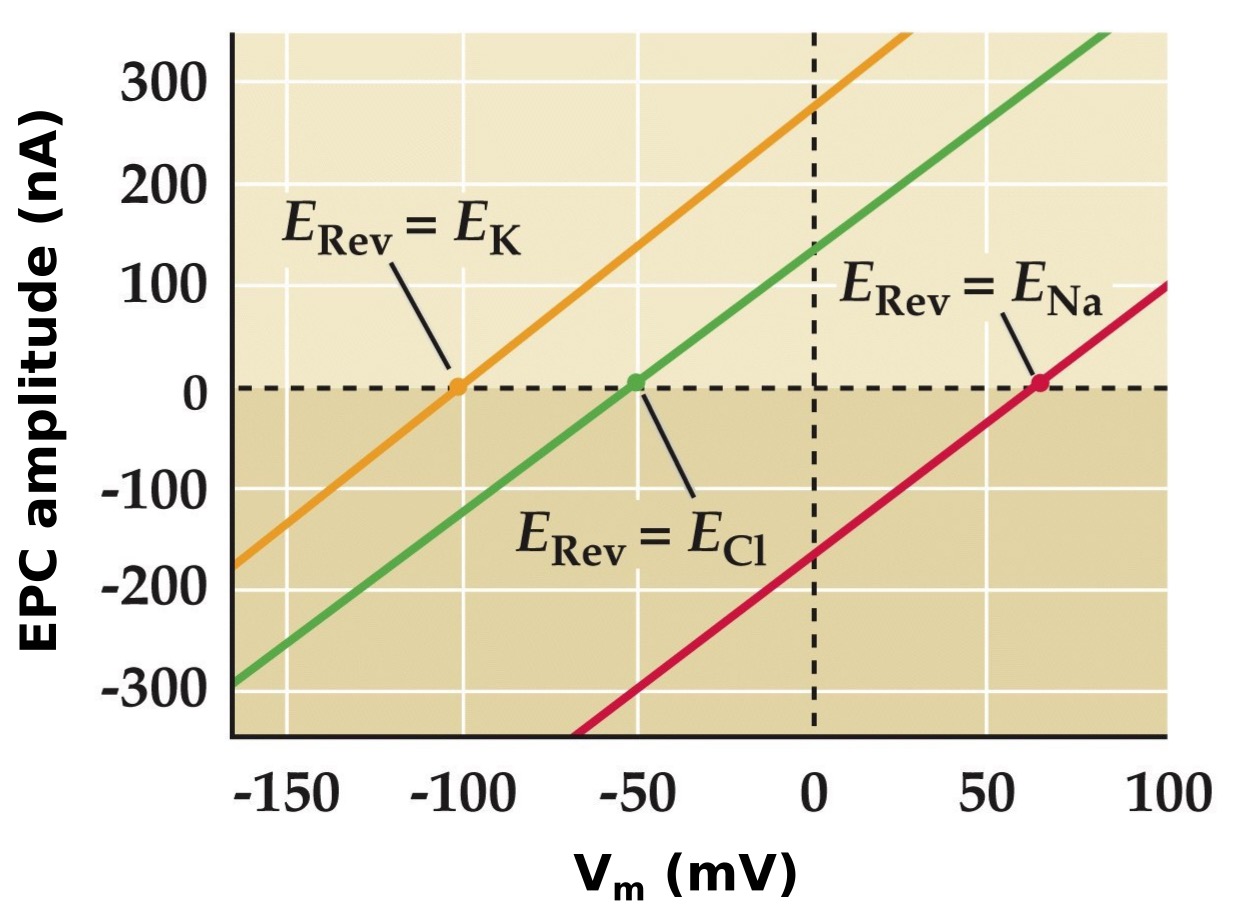
Hypothetical ion channel selectivities and the reversal potential
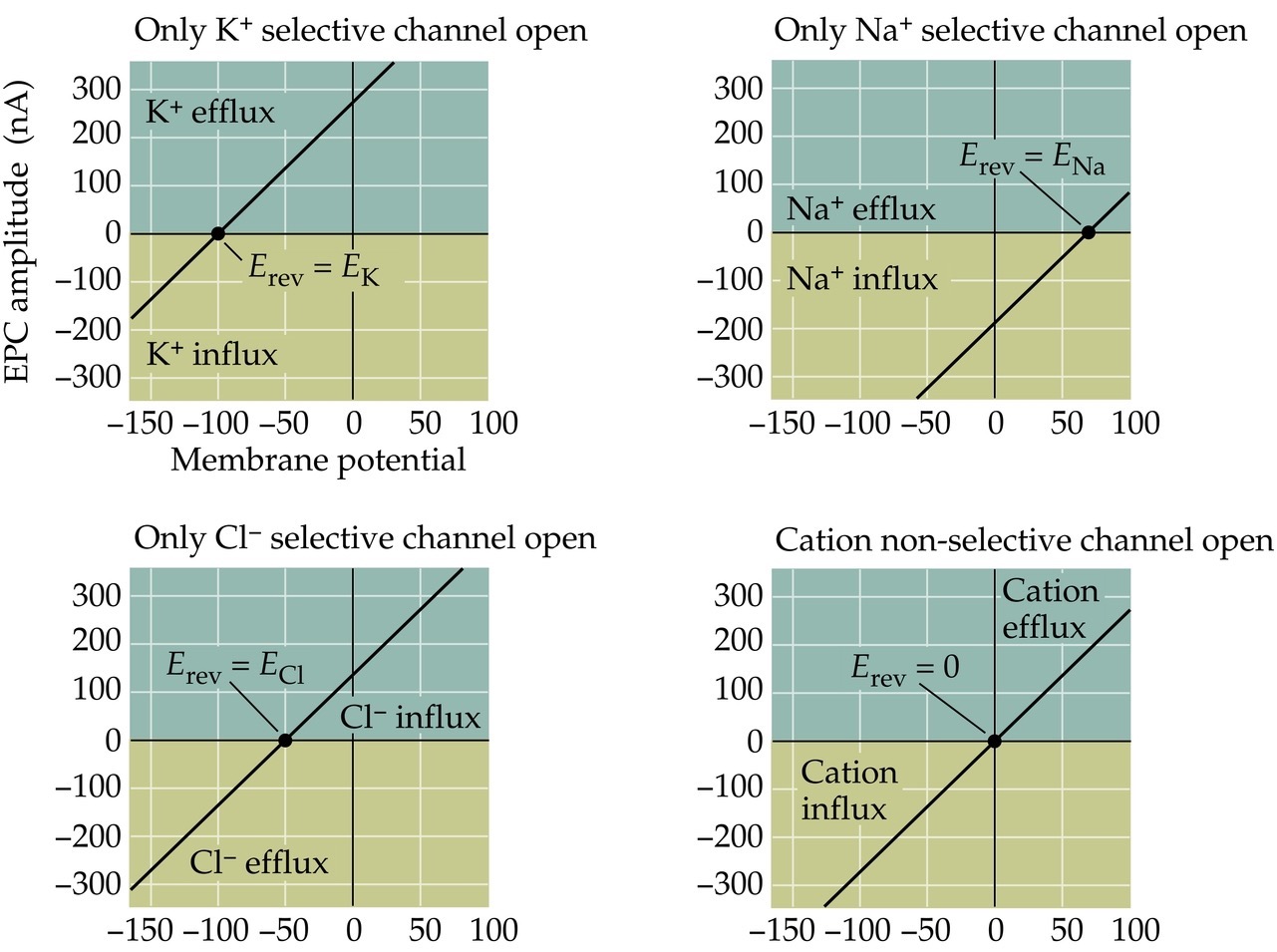
Note:
So let’s imaging what the current-voltage relationships would look like for different channel selectivities. Remember the reversal potential is when there there is no net ion flux, so it 0 nA on all these graphs and if a channel is selective to only K, it would be equal to the Ek.
If the channel was selective only to Na, than the Erev would be equal to ENa. Same for chloride.
If the channel was a non-selective cation channel (passing both K and Na) then the current-voltage relationship would look like...
11Na, 12Mg, 17Cl, 19K, 20Ca
Ca2+ ions flow through CaV channels at a rate of ~106 ions s−1, but Na+ conductance is 500fold less through CaV channels [#Tang:2014] extracellular [Na+] is nearly 70fold higher than Ca2+, thus Ca2+ selectivity is crucial [#Tang:2014] Ca2+ and Na+ have nearly identical diameters (~2 Å) 1 Å = 100 pm (Ca2+ larger atomic size, but Na+ has larger ionic size|hydration shell). Ca2+ selectivity is from high affinity binding, preventing Na+ permeability. Multi site pore, with knock on mechanism to push Ca2+ ions through [#Tang:2014]
[#Tang:2014]: Tang, L., Gamal El-Din, T. M., Payandeh, J., Martinez, G. Q., Heard, T. M., Scheuer, T., Zheng, N., and Catterall, W. A. (2014). Structural basis for Ca2+ selectivity of a voltage-gated calcium channel, Nature, 505(7481), 56-61. PMID 24270805
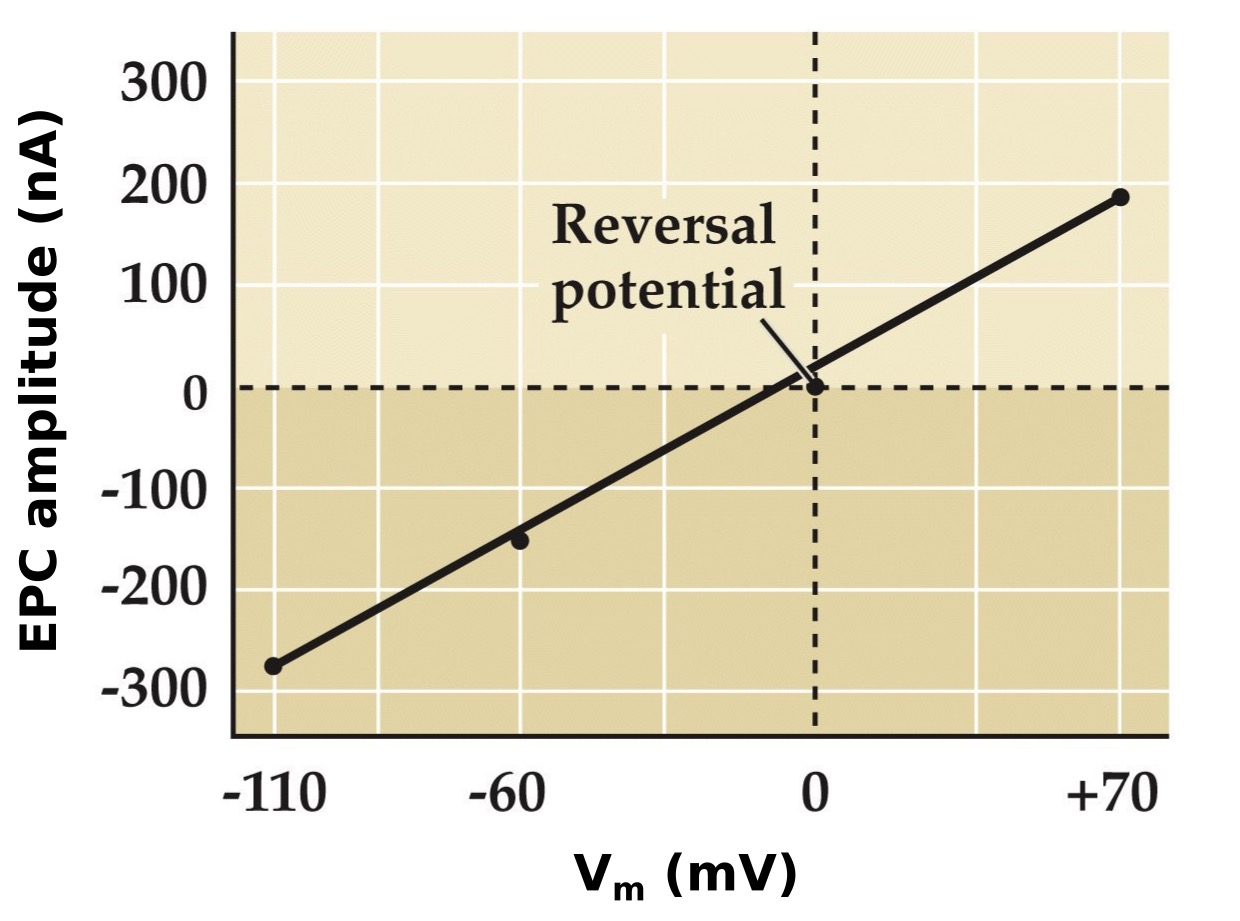
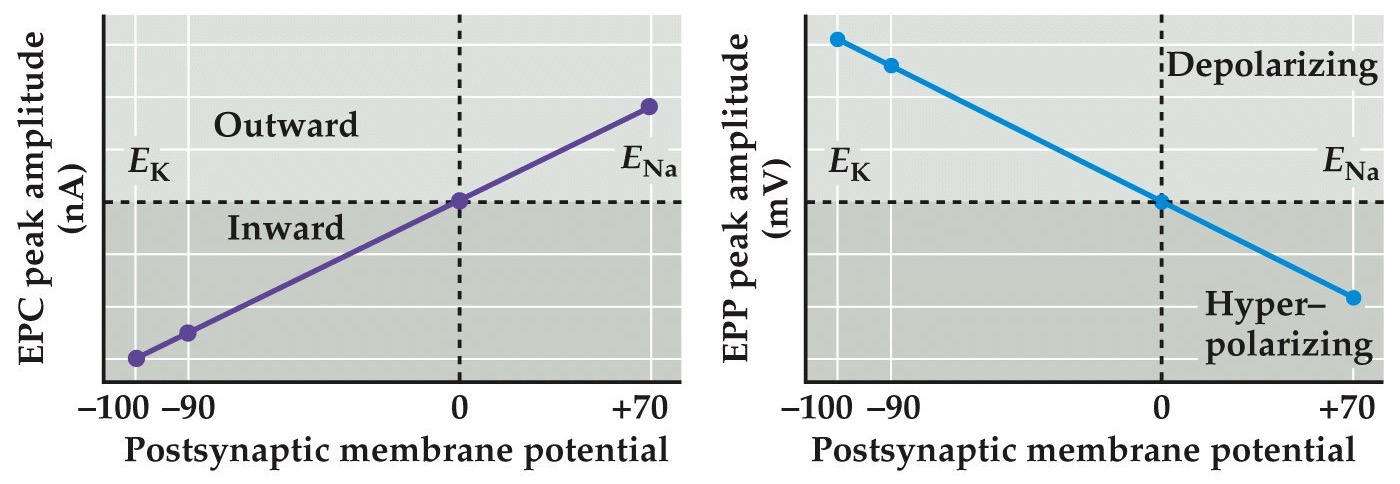
Postsynaptic Vm affects the magnitude and direction of end plate currents
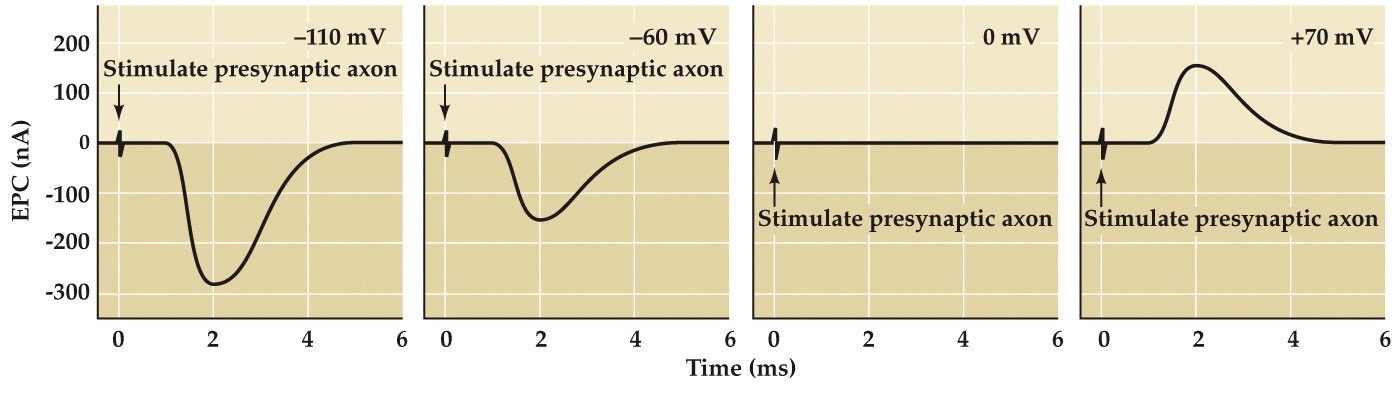
Note:
These little transients are just stimulus artifacts, but look at the postsynaptic end plate currents in these at these different Vms. Look what happens when Vm is at 0mV, there is no current and then above 0 mV it flips from being inward to net outward current...
We already know that ACh is essential for the end plate currents-- therefore we can say that this EPC is IAch. Therefore what is the Erev for IAch?
Postsynaptic Vm affects the magnitude and direction of end plate currents
Note:
[#Takeuchi:1960]: Takeuchi, A. and Takeuchi, N. (1960). On the permeability of end-plate membrane during the action of transmitter, J Physiol, 154(), 52-67. PMID 13774972
Shifting ENa+ or EK+ shifts Erev of the neuromuscular endplate current
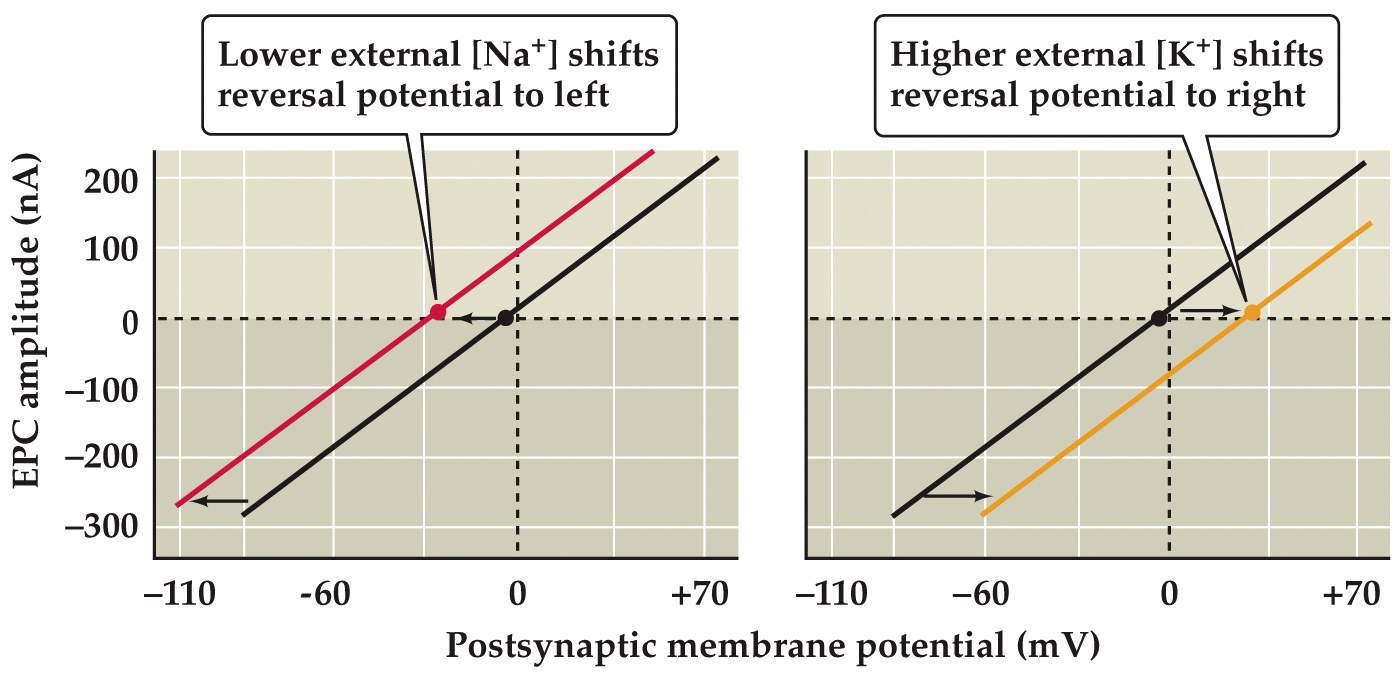
Note:
So it seems that the ACh activated ion channels are equally permeable to Na and K and this was tested in 1960 by Akira and Noriko Takeuchi by changing the extracellular concentration of these ions. As predicted, lowering [Na] shifts Erev to the left and and raising the external [K] shifts Erev to the right.
What ions flow through the nACh receptor?
- Voltage clamping experiments show that there are large inward currents at -110 mV, smaller currents at -60 mV and no current at 0 mV. Outward currents at +70 mV. Therefore Erev = 0
- Erev is not at any of the equilibrium potentials for a single ion, lies in between K⁺ (-100 mV) and Na⁺ (+70 mV)
- Altering the K⁺ concentration or the Na⁺ concentration will change the membrane potential. Therefore both Na⁺ and K⁺ are permeable through the nACh receptor
- nACh receptor can conduct both Na⁺ and K⁺ ions. The direction of flow is dependent on the membrane potential. The normal resting state of muscle is -100 mV, well below 0 mV (Erev) therefore normally at rest Na⁺ rushes in with very little K⁺ rushing out
Note:
As we will see in a minute voltage clamp experiments show that there is a…
Erev…
Furthermore, altering…
Therefore we can conclude that the nAChR can conduct both Na and K ions.
Na⁺ and K⁺ movements during EPCs and EPPs
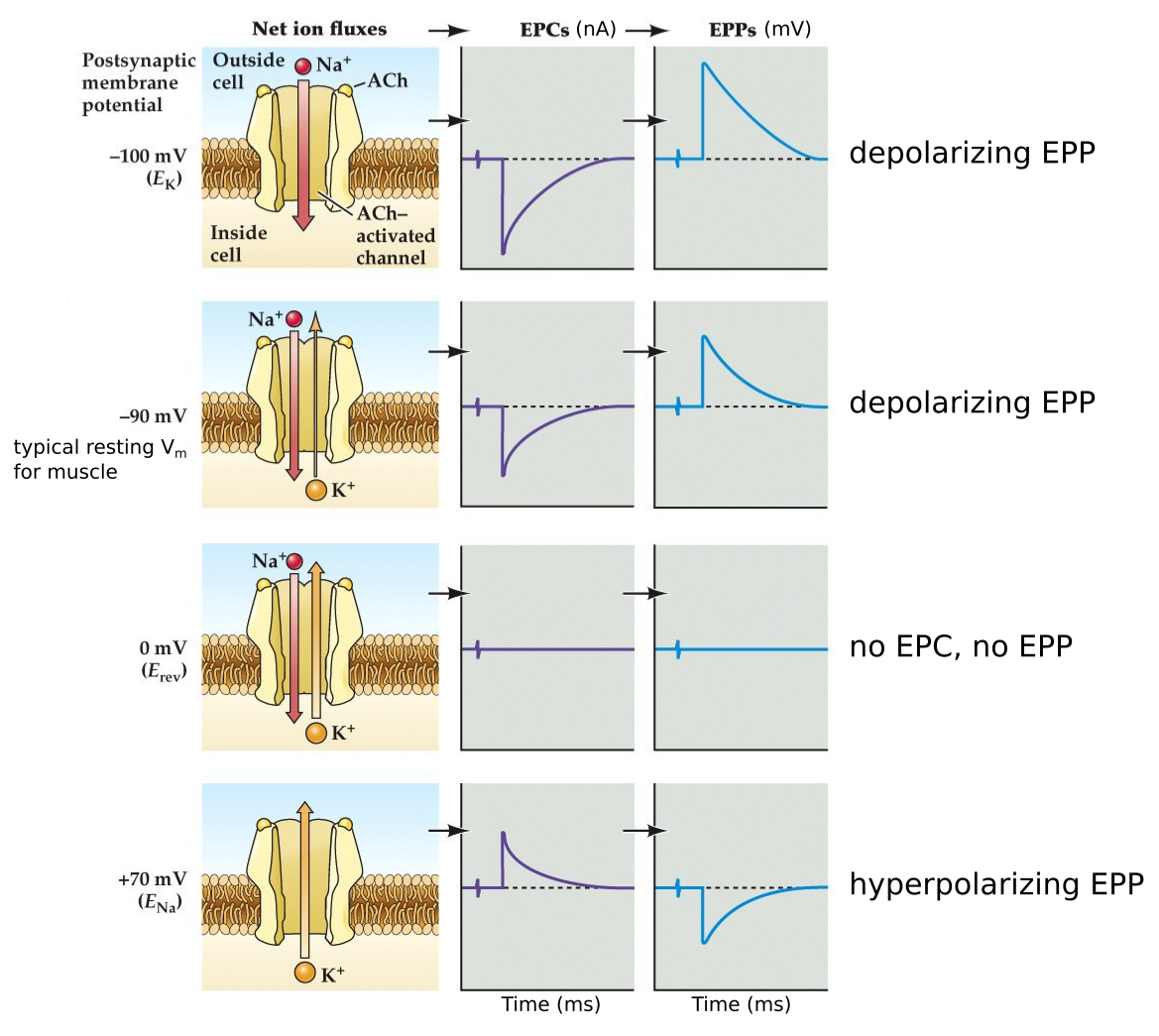
Note:
Even though these ionotropic channels opened by ACh are permeable to both Na and K, at the resting membrane potential the EPC is generated primarily by Na influx because of the reduced driving force on K since at Vrest the membrane potential is closer to Ek.
In fact the Na⁺ and K⁺ permeabilities of the nAChR channel are similar, therefore the magnitudes of the Na⁺ and K⁺ currents depends on the driving forces present for each ion
--
Na⁺ and K⁺ movements during EPCs and EPPs
Note:
Here is the key: you get inward currents at potentials more negative the Erev and you get outward currents at potentials more positive than Erev.
The resulting EPPs depolarize postsynaptic cell at potentials more negative than Erev and potentials more positive than Erev hyperpolarize the cell.
Since the Na⁺ and K⁺ permeabilities of this channel are similar, the magnitudes of the Na⁺ and K⁺ currents depends on the driving forces present for each ion
nAChR summary
- When the nAChR opens at normal resting potentials many Na⁺ ions rush in and a few K⁺ rush out. This causes a depolarizing EPP in the muscle cell. As the Vm during the EPP approaches Erev, outward K⁺ flux is equal to inward Na⁺ flux. Therefore if the nACh receptor is open long enough, it will drive Vm to Erev.
- If Erev is above action potential threshold, the probability of an action potential occurring is increased
- If Erev is below action potential threshold, the probability of an action potential occurring decreased
Note:
http://www.nature.com/nrd/journal/v1/n6/full/nrd821.html:
In the case of this modified muscle nAChR, the conductance of the pore is sensitive to the presence of negative charge at three locations that form three negatively charged rings in and near the M2 domain56. So, intensive studies of the M2 segment have been carried out to determine the amino acids that are responsible for the cationic or anionic selectivity of receptors.
Similar mechanisms exist at all chemical synapses
For synapses between neurons:
- Postsynaptic current (PSC) is similar to an end plate current
- Post synaptic potential (PSP) is similar to an end plate potential
- Excitatory PSP (EPSP)– increases likelihood of an action potential occurring
- Inhibitory PSP (IPSP)– decreases likelihood of an action potential occurring
Note:
So now let's generalize the properties that we’ve learned about EPCs through ionotropic AChR and their effects on EPPs at the neuromuscular junction to the case of chemical synapses between any pair of neurons...
But instead of the so called EPPs, we'll call the postsynaptic potentials between neurons we call excitatory PSP if it increases the likelihood of an AP firing in a postsynaptic cell and inhibitory PSP if it decr the probability of an AP occurring in a postsynaptic cell.
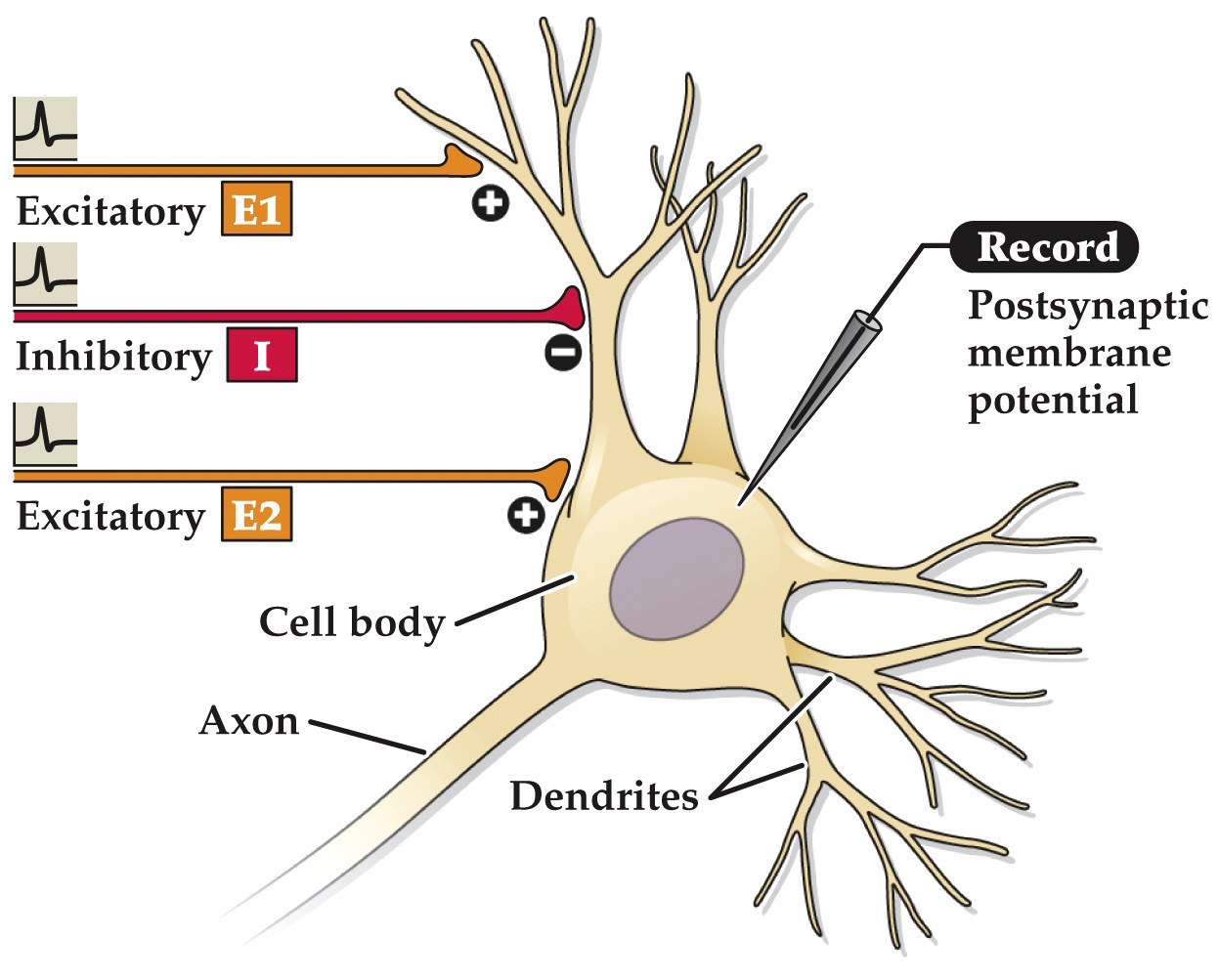
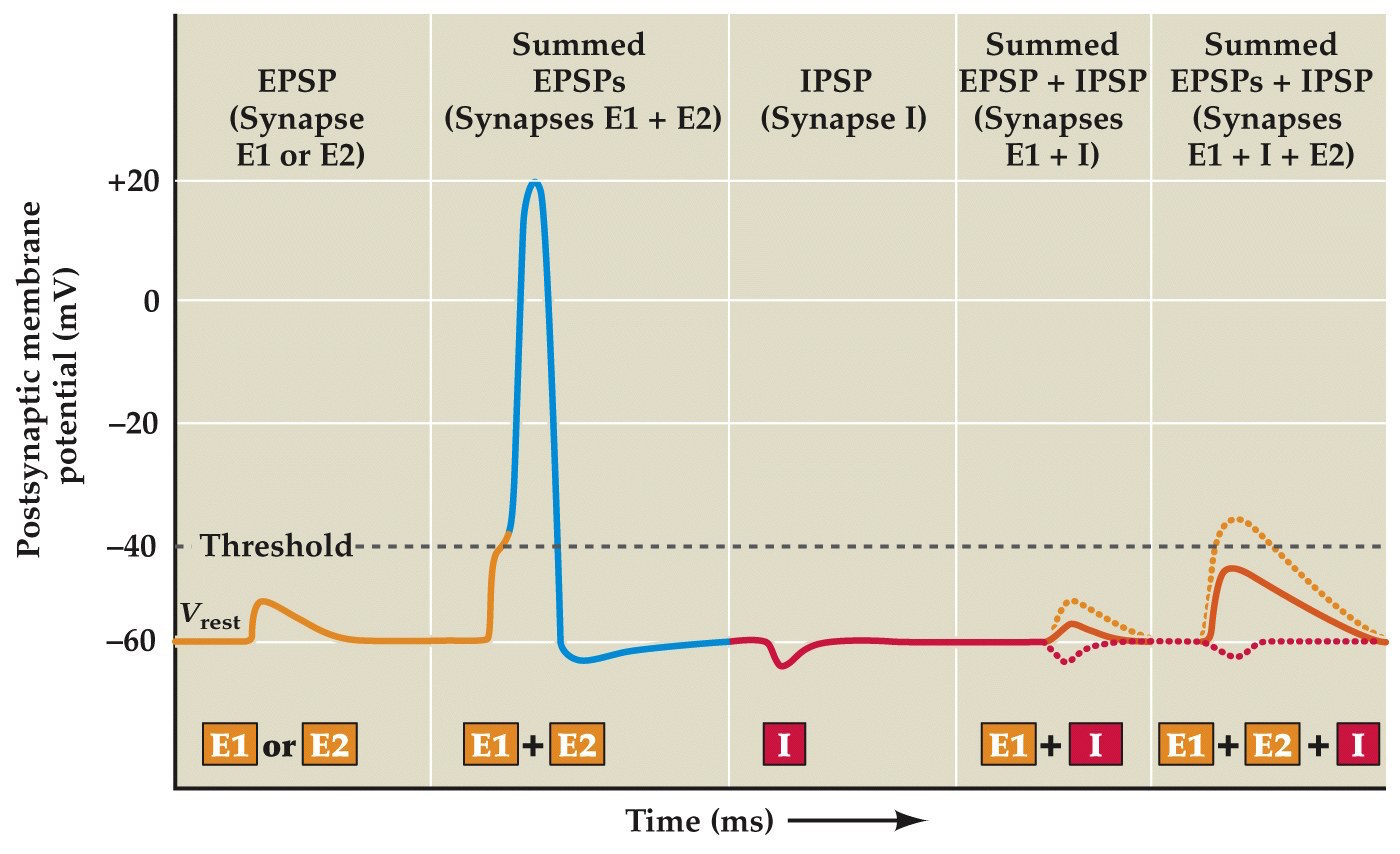
EPSP summation
- Unlike the neuromuscular junction– at synapses between neurons an individual EPSP is usually not very strong, typically well below threshold.
- Multiple EPSPs need to be summed together for the neuron's Vm to reach threshold. Individual neurons can receive thousands synapses. It's the summation of EPSPs and IPSPs that determine whether or not an action potential occurs.
Note:
Excitatory postsynaptic potential (EPSP)
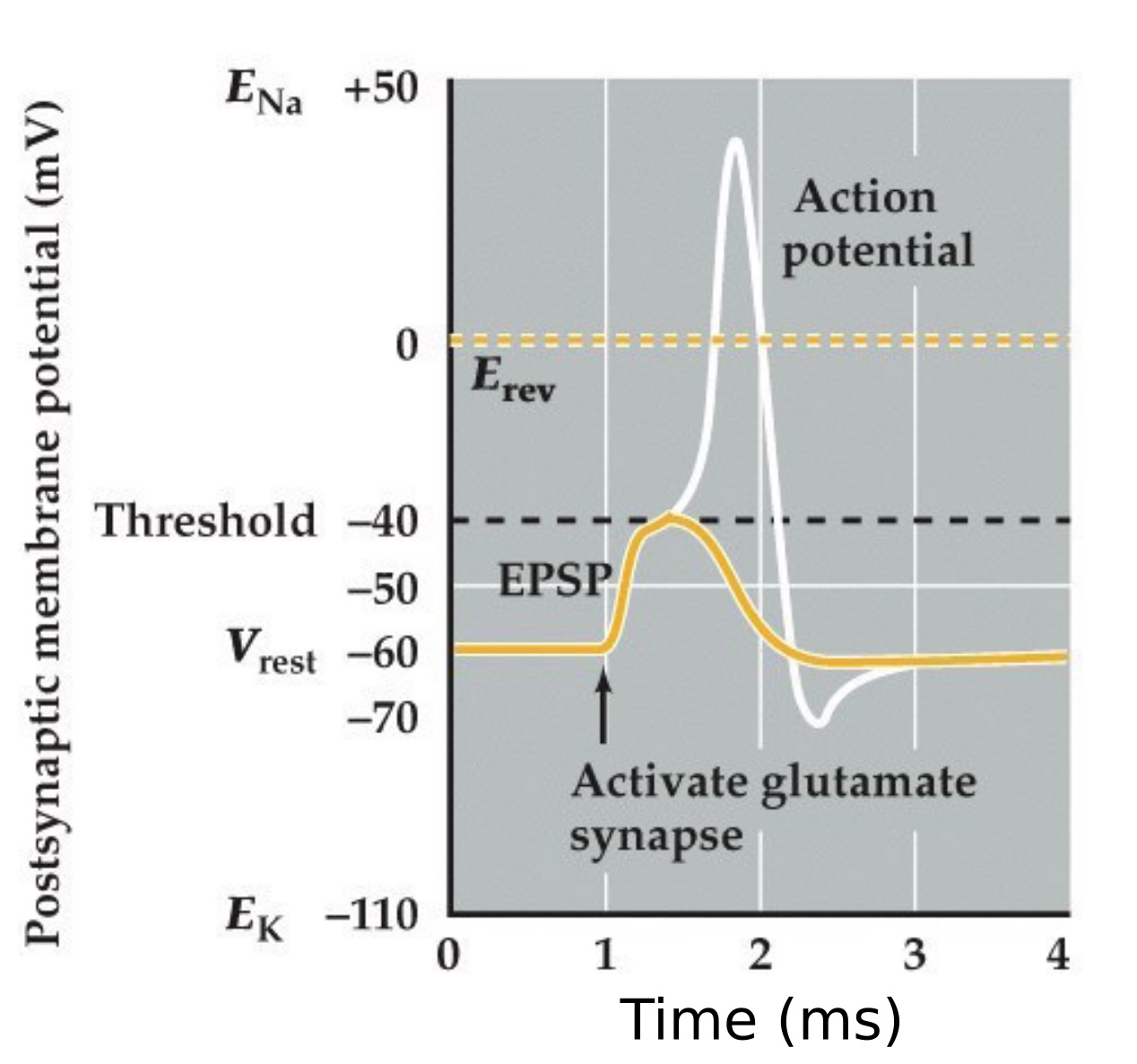
Note:
Imagine an experiment like the endplate potental recordings at the neuromuscular junction before but this time on a neuron in the CNS
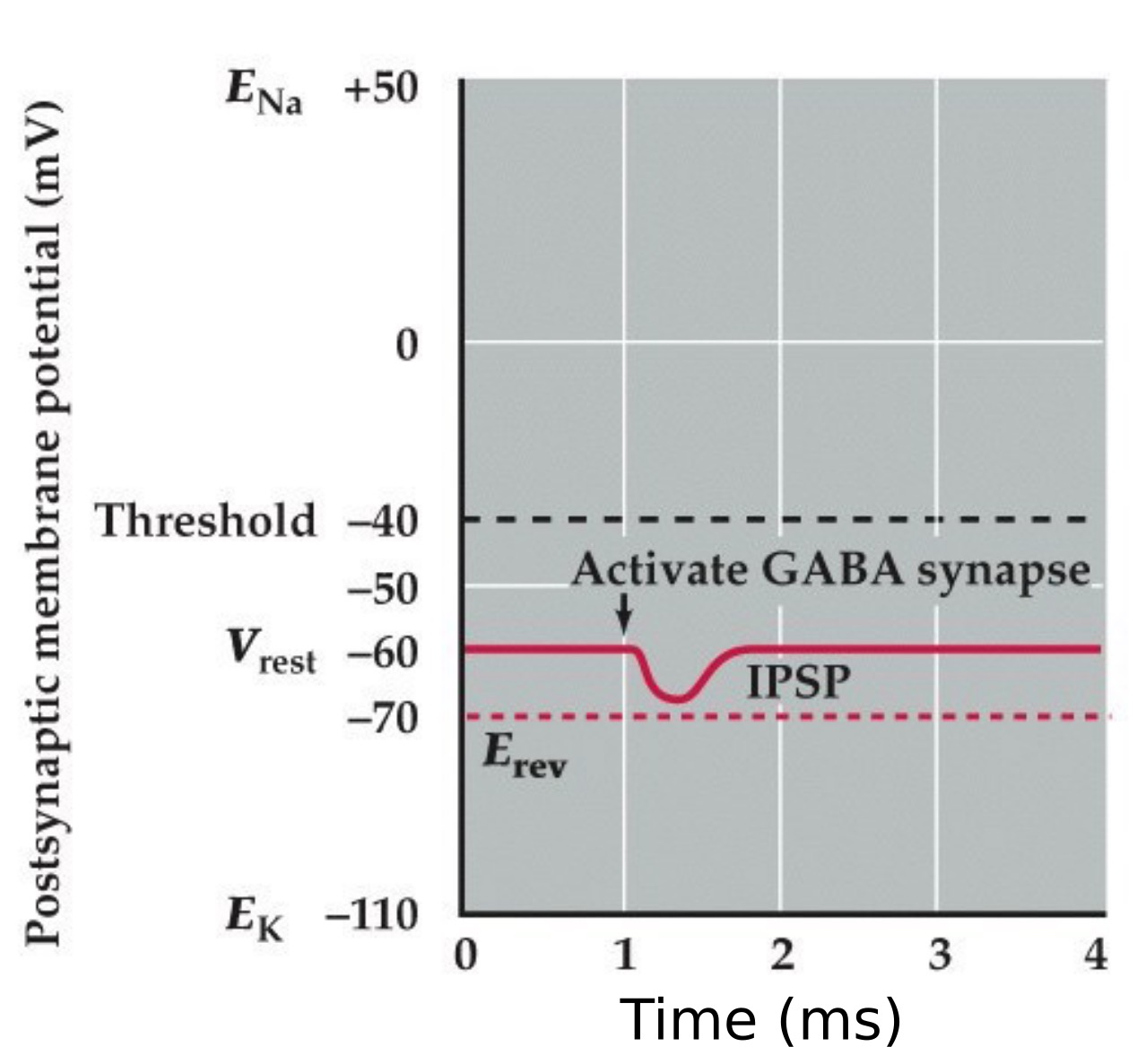
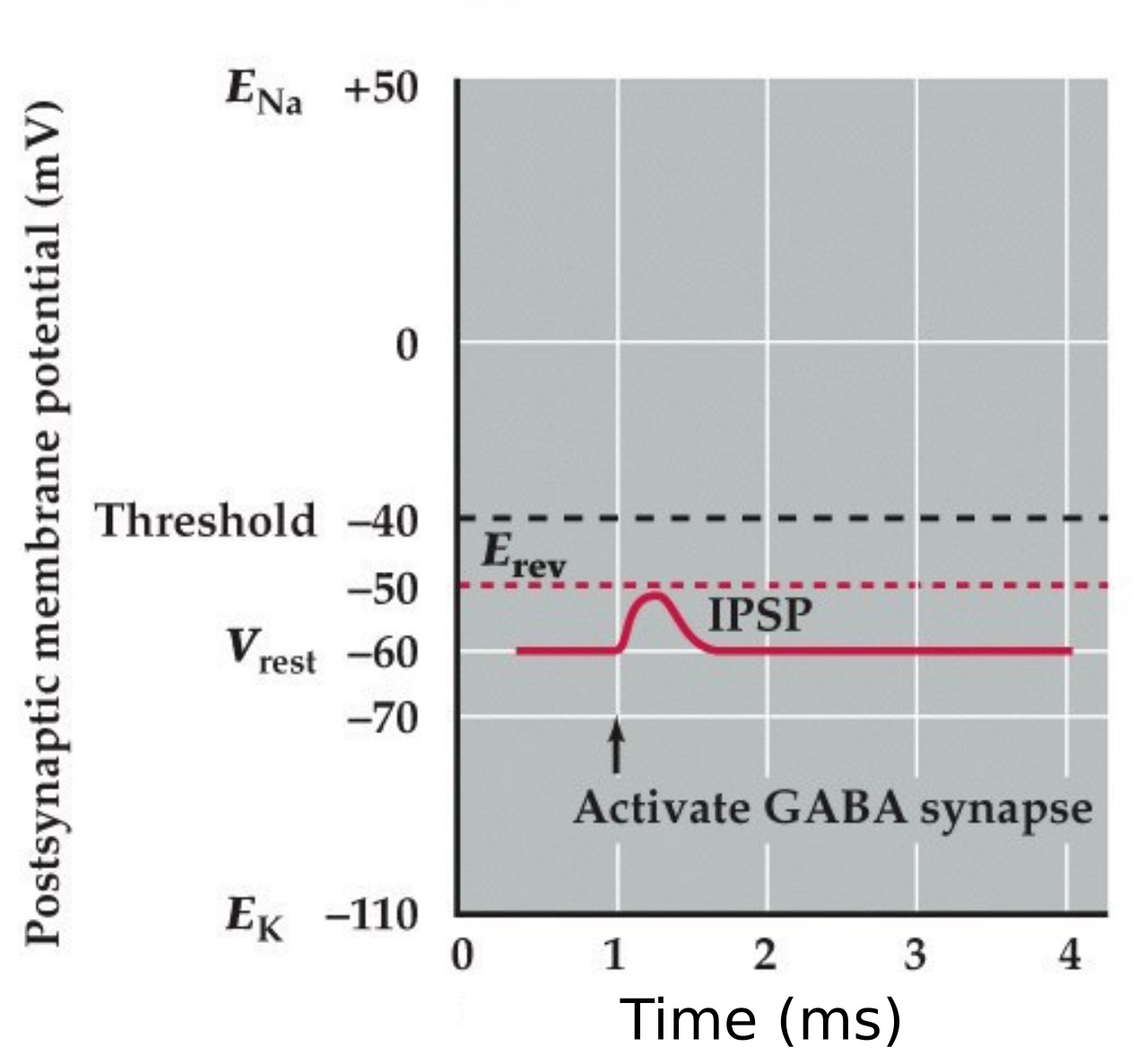
Inhibitory postsynaptic potential (IPSP) type 1
- An IPSP mediated by a GABA activated chloride selective channel that hyperpolarizes the neuron
- Reversal potential for the Cl⁻ current is negative to the resting potential and action potential threshold
Note:
IPSP type 2
- The reversal potential for the Cl⁻ current is positive to the resting potential but negative to threshold
- Activation of Cl⁻ channels depolarizes the neuron. Stabilizes membrane potential below threshold
Note:
Imagine if a separate EPSP input brought Vm of this neuron to -41 mV, just below -40mV threshold. Since this is now postive to the ECl of -50mV, further activity at the IPSP synapses will now hyperpolarize the neuron back towards -50mV.
This can also be called shunting inhibition. In this case Na⁺ channels could persistently be in a state of inactivation due to small ongoing depolarizing and hyperpolarzing pulses keeping the neurons Vm below threshold.
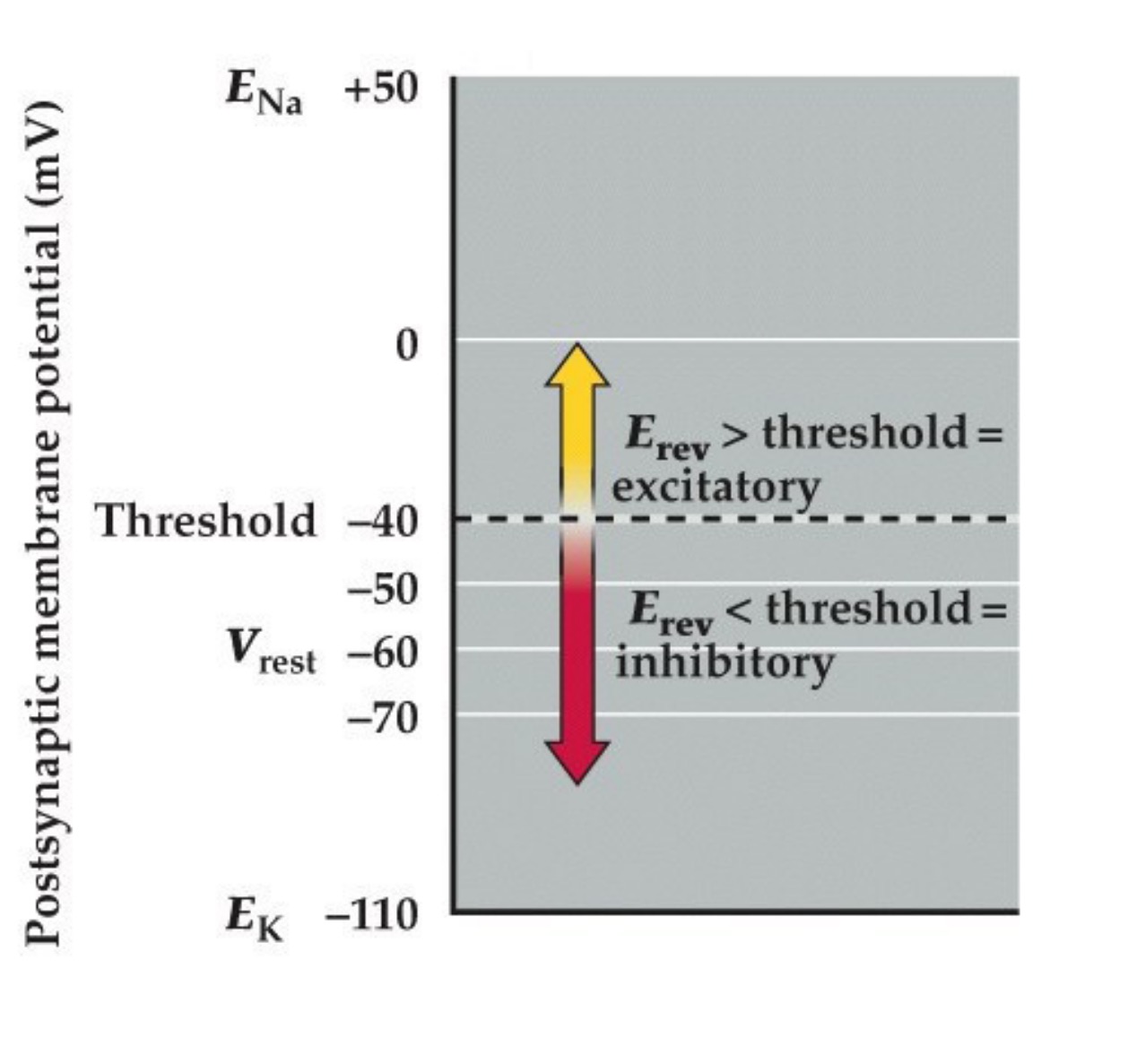
So just remember, the key is that if the Erev for the neurotransmitter receptor is more positive than threshold than it is excitatory. If it is more negative than threshold than it is inhibitory.
Blocking NKCC1 with bumetanide disrupts excitatory synapse development in the cortex
Bumetanide, a selective NKCC1 inhibitor, has been demonstrated to suppress certain forms of epileptiform activity in vitro and in vivo, presumably by attenuating the depolarizing effect of GABA (Dzhala et al., 2005; Kilb et al., 2007)
effect of GABA on membrane polarity depends on the Cl gradient created by the expression of Na -K -2Cl cotransporter (NKCC) and K-Cl cotransporter (KCC). NKCC1 imports Cl and is expressed from the embryonic stage until the first postnatal week, whereas KCC2 exports Cl and is weakly expressed at birth and upregulated as the brain matures (Plotkin et al., 1997; Rivera et al., 1999; Li et al., 2002). The temporal expression patterns of these two transporters correspond to the switch of GABA from being excitatory to inhibitory during the first few weeks of rodent postnatal life (Delpire, 2000).
Summation of postsynaptic potentials
Note:
Summation
- In general EPSPs in neurons are small 0.2–0.4 mV
- Most neurons are somewhere between 10–20 mV below threshold. If everything was linear that it would take the sum of 50 or so inputs to trigger AP
- Not so simple-- synaptic inputs can be summed in space and time within a neuron
- Recall a single neuron may have as many as 10,000 different synapses. Some are excitatory some inhibitory, some strong some weak. Some at the tips of dendrites, some near the cell body
- Integration of all these little postsynaptic bioelectric waves determines whether the neuron fires an action potential
Note:
Neural integration
- How does a neuron integrate all the information it is getting?
- For many neurons the axon hillock is where the decision to initiate an action potential is made. Contains a high density of voltage dependent Na^+^ channels; membrane with the lowest threshold
- Local state of the cell is sensed at the axon hillock. The bioelectrical state of the cell is a function of time, the combination of all the EPSPs and IPSPs going on at any moment
- The combined sub- or supra-threshold synaptic potential waveforms spread passively to the soma and axon hillock
- Temporal summation-- consecutive signals within a period of time are added together
- Spatial structure of the dendrites determines the degree by which a synaptic potential current decreases as it spreads passively. Summation of inputs is easier on same dendritic branch than on different branches
Note:
- Different synapses will have different time constants
- Some dendrites have voltage gated Na^+^ channels (albeit lower density than axons), these can amplify inputs
- Length constant of the cell determines the degree to which a depolarization current decreases as it spreads passively. Easier to sum inputs on the same dendritic branch than on different branches
- Time constant
- time needed for for resistive current (I
r, current due to ions flowing through channels) and membrane potential (Vm) to reach 63% of their asymptotic values is proportional to the combination of resistance and capacitance of the circuit in question (across the cell membrane) - membrane current (I
m) is sum of Irand the capacitive current (Ic) - I
m= Ir+ Ic - capacitance of membrane: during change in applied voltage or current across membrane, positively charged ions pile on surface of one side of membrane and electrostatically interact with cations on the other side of membrane surface (membrane acts as thin impermeable surfaces in parallel, like a capacitor), repeling them and inducing immediate, fast capacitive current along membrane
- capacitive current falls with an exponential time course. And the membrane potential rises with same exponential time course
- Relation of membrane potential at time t during charging of capacitance is given by V
t= Vinf(1 - e^-t/RC^), where Vinfis the membrane potential at an infinite asymptotic value of the exponential curve. When t = RC, then we have Vt= Vinf( 1 - e^-1^) ==> Vinf(0.63)
console.log( 1 - Math.E ** -1)